UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
WASHINGTON, D.C. 20549
FORM 10-K
| |
|
|
| þ |
|
ANNUAL REPORT PURSUANT TO SECTION 13 OR 15 (d) OF
THE SECURITIES EXCHANGE ACT OF 1934 |
For the fiscal year ended December 31, 2009
Or
| |
|
|
| o |
|
TRANSITION REPORT PURSUANT TO SECTION 13 OR 15 (d) OF THE
SECURITIES EXCHANGE ACT OF 1934 |
Commission File Number 000-21243
OncoGenex Pharmaceuticals, Inc.
(Exact name of the registrant as specified in its charter)
| |
|
|
|
Delaware
|
|
95-4343413 |
(State or other jurisdiction of
incorporation or organization)
|
|
(I.R.S. Employer
Identification No.) |
1522 217 th Place SE, Suite 100, Bothell, Washington 98021
(Address of principal executive offices, including zip code)
(425) 686-1500
(Registrants telephone number, including area code)
Securities registered pursuant to Section 12(b) of the Act:
| |
|
|
|
Title of Each Class
|
|
Name of Exchange on Which Registered |
|
|
|
|
| Common Stock, par value $0.001 per share
|
|
The NASDAQ Capital Market |
Securities registered pursuant to Section 12(g) of the Act:
None
Indicate by check mark if the registrant is a well-known seasoned issuer,
as defined in Rule 405 of the Securities Act. Yes o No þ
Indicate by check mark if the registrant is not required to file reports
pursuant to Section 13 of 15(d) of the Act. Yes o No þ
Indicate by check mark whether the registrant: (1) has filed all reports
required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934
during the preceding 12 months (or for such shorter period that the registrant was
required to file such report), and (2) has been subject to such filing requirements for
the past 90 days. Yes þ No o
Indicate by check mark whether the registrant has submitted electronically and
posted on its corporate Web site, if any, every Interactive Data File required to be
submitted and posted pursuant to Rule 405 of Regulation S-T (§ 232.405 of this chapter)
during the preceding 12 months (or for such shorter period that the registrant was
required to submit and post such files). Yes o No o
Indicate by check mark if disclosure of delinquent filers pursuant to
Item 405 of Regulation S-K is not contained herein, and will not be contained, to the
best of the registrants knowledge, in definitive proxy or information statements
incorporated by reference in Part III of this Form 10-K or any amendment to this
Form 10-K. þ
Indicate by check mark whether the registrant is a large accelerated
filer, an accelerated filer, a non-accelerated filer, or a smaller reporting company.
See the definitions of large accelerated filer, accelerated filer, and smaller
reporting company in Rule 12b-2 of the Exchange Act.
| |
|
|
|
|
|
|
| Large accelerated filer o
|
|
Accelerated filer þ
|
|
Non-accelerated filer o
|
|
Smaller reporting company o |
Indicate by check mark whether the registrant is a shell company (as
defined in Rule 12b-2 of the Exchange Act.). Yes o No þ
As of June 30, 2009, the aggregate market value of the registrants
Common Stock held by non-affiliates of the registrant was $97,175,075. As of March 4,
2010, 6,333,685 shares of the registrants Common Stock were outstanding.
DOCUMENTS INCORPORATED BY REFERENCE
Portions of the registrants definitive Proxy Statement to be
filed with the Securities and Exchange Commission not later than April 30, 2009, in
connection with the solicitation of proxies for its 2010 Annual Meeting of
Shareholders, are incorporated by reference into Items 10, 11, 12, 13 and 14 of Part
III hereof.
OncoGenex Pharmaceuticals, Inc.
Table of Contents
2
PART I
References in this Form 10-K to OncoGenex Pharmaceuticals, OncoGenex, the Company, we, us
or our refer to OncoGenex Pharmaceuticals, Inc. and its wholly owned subsidiaries. The
information in this Form 10-K contains certain forward-looking statements, including statements
related to clinical trials, regulatory approvals, markets for the Companys products, new product
development, capital requirements and trends in its business that involve risks and uncertainties.
The Companys actual results may differ materially from the results discussed in the
forward-looking statements. Factors that might cause such a difference include those discussed in
Business, Risk Factors and Managements Discussion and Analysis of Financial Condition and
Results of Operations as well as those discussed elsewhere in this Form 10-K.
ITEM 1. BUSINESS
Overview of Our Business
OncoGenex is a biopharmaceutical company committed to the development and commercialization of new
cancer therapies that address unmet needs in the treatment of cancer. OncoGenex has five product
candidates in its pipeline, namely, OGX-011, OGX-427, OGX-225, SN2310 and CSP-9222, with each
product candidate having a distinct mechanism of action and representing a unique opportunity for
cancer drug development.
Product Candidates Overview and Recent Developments
Our product candidates OGX-011, OGX-427 and OGX-225 focus on mechanisms of treatment resistance in
cancer patients and are designed to address treatment resistance by blocking the production of
specific proteins which we believe promote survival of tumor cells and are over-produced in
response to a variety of cancer treatments. Our aim in targeting these particular proteins is to
disable the tumor cells adaptive defenses and thereby render the tumor cells more susceptible to
attack with a variety of cancer therapies, including chemotherapy, which we believe will increase
survival time and improve the quality of life for cancer patients. Product candidate SN2310 is a
novel camptothecin for the treatment of cancer. Camptothecins are potent anticancer agents that
belong to the family of drugs called topoisomerase I inhibitors that bind reversibly to the
TOPO-I-DNA complex causing breaks in the DNA strands during replication resulting in cell death.
Product candidate CSP-9222 is the lead compound from a family of compounds, which have been
in-licensed from Bayer Healthcare LLC (Bayer), that demonstrates activation of programmed cell
death in pre-clinical models.
OGX-011
In December 2009, we announced that our wholly-owned subsidiary, OncoGenex Technologies Inc.
(OncoGenex Technologies), entered into a Collaboration and License Agreement (Collaboration
Agreement) with Teva Pharmaceutical Industries Ltd. (Teva), for the development and global
commercialization of OGX-011 (and related compounds targeting clusterin, excluding OGX-427 and
OGX-225). In connection with the Collaboration Agreement, OncoGenex and Teva developed a Clinical
Development Plan (the Clinical Development Plan) under which the following three phase 3 clinical
trials will be initiated:
| |
|
|
a phase 3 clinical trial of OGX-011 in approximately 300 patients with second-line
castrate resistant prostate cancer (CRPC), expected to initiate in the second quater of 2010 subject to institutional review board approval and patient screening. OncoGenex will
have primary responsibility for the oversight of this trial; |
| |
|
|
a phase 3 clinical trial of OGX-011 in approximately 800 patients with first-line CRPC,
expected to initiate in the third quater of 2010. Teva will have primary responsibility for the oversight of
this trial; and |
| |
|
|
a phase 3 clinical trial of OGX-011 in at least 700 patients with first-line non-small
cell lung cancer (NSCLC), expected to initiate by early 2011. Teva will have primary
responsibility for the oversight of this trial. |
3
Under the Collaboration Agreement, Teva agreed to pay us upfront payments in the aggregate amount
of $50 million, will pay up to $370 million upon the achievement of developmental and commercial
milestones and will pay royalties at percentage rates ranging from the mid-teens to mid-twenties on
net sales. We have an option to co-promote OGX-011 in the United States and Canada.
In connection with the Collaboration Agreement, the Company and Teva also entered into a stock
purchase agreement (the Stock Purchase Agreement) pursuant to which Teva made an additional $10
million equity investment in OncoGenex at a 20% premium to a thirty-day average closing share
price.
Funding responsibilities for the Clinical Development Plan have been allocated as follows:
| |
|
|
We are required to contribute $30 million towards development of OGX-011 which will
include our personnel costs for certain development activities; and |
| |
|
|
Teva is required to fund all other expenses under the Clinical Development Plan. |
In addition to the development costs noted above, Teva is also responsible for all costs relating
to product commercialization, including any costs incurred in relation to our co-promotion option,
except for start-up costs of any exercised co-promotion activities in advance of commercialization.
For a detailed discussion of the terms of the Collaboration Agreement, refer to the discussion
under the heading License and Collaboration Agreements Teva Pharmaceutical Industries Ltd.
OncoGenex has conducted five phase 2 clinical trials to evaluate the ability of OGX-011 to enhance
the effects of therapy in prostate, non-small cell lung and breast cancers. Results have been
presented for each of these phase 2 studies. OncoGenex has designed phase 3 registration clinical
trials to evaluate the clinical benefit of OGX-011 in first and second-line CRPC. Together with
Teva, we are in the process of designing a phase 3 registration trial with respect to NSCLC. Refer
to the discussion below under the headings Summary of OGX-011 Product Registration Strategy and
Summary of Results of OGX-011 Phase 2 Clinical Trials for further details.
OGX-427
OGX-427, an inhibitor of heat shock protein 27, is being evaluated in a phase 1 clinical trial to
evaluate safety for OGX-427 administered alone, as well as in combination with docetaxel
chemotherapy (docetaxel), in patients with various types of cancer. Enrollment in the OGX-427
monotherapy aspect of the phase 1 clinical trial is complete. Enrollment in the OGX-427 in
combination with docetaxel aspect of the clinical trial is ongoing. A second investigator-sponsored
phase 1 clinical trial evaluating OGX-427 when administered directly into the bladder in patients
with bladder cancer was initiated in August 2009. The study, which will enroll up to 36 patients
with bladder cancer, is designed to determine the safety and potential benefit of OGX-427
administered directly into the bladder using a catheter, which is known as intravesical
instillation. In addition, the study will measure the direct effect of OGX-427 on expression of
Hsp27 in bladder tumor cells as well as determine the pharmacokinetics and pharmacodynamics of
OGX-427 when delivered by intravesical instillation.
In January 2010, we announced that an investigator-sponsored phase 2 clinical trial evaluating
OGX-427 when administered as a monotherapy to patients with CRPC has received grant funding. The
randomized, controlled phase 2 study will enroll up to 72 patients and is designed to determine the
potential benefit of OGX-427 by evaluating the number of patients who are without disease
progression at 12 weeks post-study treatment, with or without OGX-427.
4
SN2310, OGX-225 and CSP-9222
SN2310 was evaluated in a phase 1 clinical trial to evaluate safety in patients with advanced
cancer who have received on average 3 to 5 prior chemotherapy treatments. The phase 1 clinical
trial has been completed. We are currently exploring options to out-license this product candidate.
OGX-225 is a second generation antisense drug which in preclinical experiments, inhibits production
of both Insulin Growth Factor Binding Protein 2 (IGFBP-2) and Insulin Growth Factor Binding
Protein-5 (IGFBP-5). Increased IGFBP-2 or IGFBP-5 production is observed in many human cancers,
including prostate, non-small cell lung, breast, ovarian, bladder, pancreatic, colon, acute myeloid
leukemia, acute lymphoblastic leukemia, neuroblastoma, glioma and melanoma cancers. Increased
IGFBP-2 or IGFBP-5 production is linked to faster rates of cancer progression, treatment resistance
and shorter survival duration in humans.
Product candidate CSP-9222, in
pre-clinical development, is the lead compound from a family of compounds, which have been
in-licensed from Bayer Healthcare LLC (Bayer), that demonstrates activation of programmed
cell death in pre-clinical models.
Financial Overview
In the fourth quarter of 2009, we recognized $25.5 million in collaboration revenue attributable to
our upfront payment from Teva. Previously, we had not recognized any revenue. We have devoted
substantially all of our resources to the development of our product candidates.
We incurred a loss for the year ended December 31, 2009 of $5.5 million and had a cumulative loss
at December 31, 2009 of $53.5 million and expect to continue to incur increasing additional losses
in the future as we continue our research and development activities.
To date, we have funded our operations primarily through the private and public placements of
equity securities and upfront payments received from our Collaboration Agreement with Teva.
Management believes that additional human resources will be required to carry on existing
development activities, though some of these costs will be passed through to Teva. We are unable to
predict when, if ever, we or Teva will be able to commence the sale of OGX-011 or any of our other
product candidates.
Recent Corporate History
On August 21, 2008, the Company, then named Sonus Pharmaceuticals, Inc., completed its acquisition
(the Arrangement) of OncoGenex Technologies, a Canadian corporation, as contemplated by the
Arrangement Agreement between the companies dated May 27, 2008 (the Arrangement Agreement). As a
result of the Arrangement, OncoGenex Technologies became a wholly-owned subsidiary of the Company.
As used in this filing, the term Sonus is used to refer to the business of the Company prior to
August 21, 2008. More information concerning the Arrangement is contained in note 5 to the
financial statements included in this Annual Report on Form 10-K, as well as in our Current Report
on Form 8-K filed on August 21, 2008 and our Definitive Proxy Statement on Schedule 14A filed on
July 3, 2008.
The consolidated financial statements account for the Arrangement as a reverse acquisition, whereby
OncoGenex Technologies is deemed to be the acquiring entity from an accounting perspective. The
consolidated results of operations of the Company for the years ended December 31, 2009 and
December 31, 2008 include the results of operations of only OncoGenex Technologies for the time
period of January 1, 2008 through August 20, 2008 and include the results of the combined company
following the completion of the Arrangement on August 21, 2008. The consolidated results of
operations for the year ended December 31, 2007 include only the consolidated results of operations
of OncoGenex Technologies and do not include historical results of Sonus. This treatment and
presentation is in accordance with SFAS 141. Information relating to the number of shares, price
per share and per share amounts of common stock are presented on a post- reverse stock split basis,
as a reverse stock split in the ratio of one-for-eighteen was effected in connection with the
Arrangement.
The Company was incorporated in October 1991 and OncoGenex Technologies was incorporated in
May 2000.
Our Product Candidates
We have three clinical-stage product candidates (OGX-011, OGX-427, and SN2310) and two
pre-clinical-stage product candidates (CSP-9222 and OGX-225).
5
OGX-011
Overview of OGX-011
Through our clinical trials, we are treating cancer patients with OGX-011 to reduce clusterin
production. Clusterin is a protein that is over-produced in several types of cancer and in response
to many cancer treatments, including hormone ablation therapy, chemotherapy and radiation therapy.
Preclinical and other data suggest that clusterin promotes cell survival. Increased clusterin
production has been linked to faster rates of cancer progression, treatment resistance and shorter
survival duration. Since increased clusterin production is observed in many human cancers,
including prostate, non-small cell lung, breast, ovarian, bladder, renal, pancreatic, anaplastic
large cell lymphoma and colon cancers and melanoma, we believe that OGX-011 may have broad market
potential to treat many cancer indications and disease stages.
A broad range of pre-clinical studies conducted by the Vancouver Prostate Centre and others have shown that reducing clusterin production with
OGX-011: (i) facilitates tumor cell death by sensitizing human prostate, non-small cell lung,
breast, ovarian, bladder, renal and melanoma tumor cells to various chemotherapies; and (ii)
sensitizes prostate tumor cells to hormone ablation therapy and sensitizes prostate and non-small
cell lung tumor cells to radiation therapy. Pre-clinical studies conducted by the Vancouver Prostate Centre
also indicate that reducing clusterin production with OGX-011 re-sensitizes docetaxel-resistant
prostate tumor cells to docetaxel.
Our phase 1 clinical trials evaluated the safety and established a recommended phase 2 dose of
OGX-011 in combination with docetaxel (two different schedules), gemcitabine and a platinum
chemotherapy or hormone ablation therapy. Docetaxel, gemcitabine, and a platinum regimen are all
examples of chemotherapy agents. In all of our phase 1 clinical trials, 640 mg, the highest dose
evaluated, was well tolerated and established as the recommended phase 2 dose.
We have conducted five phase 2 clinical trials to evaluate the ability of OGX-011 to enhance the
effects of therapy in prostate, non-small cell lung and breast cancer. These phase 2 clinical
trials have treated 294 patients with OGX-011 (or over 300 patients if including patients in
control groups). Data is available from each of the five phase 2 studies which demonstrate that
adding OGX-011 to therapy shows potential benefit of OGX 011, including:
| |
|
|
longer survival duration when adding OGX-011 to first-line docetaxel compared to
first-line docetaxel alone in patients with CRPC within a randomized phase 2 trial; |
| |
|
|
longer survival duration when adding OGX-011 to either mitoxantrone or docetaxel as
second-line chemotherapy compared to survival duration observed in two published studies
of CRPC patients receiving second-line chemotherapy (One is a study in patients with
better prognostic risk factors who received docetaxel as second-line chemotherapy which
was conducted at the British Columbia Cancer Agency and presented at the American Society
of Clinical Oncology (ASCO) Genitourinary Conference in 2008 (BCCA Study) and the
second study is the follow-up evaluation of patients on the TAX 327 Study (the TAX 327
Study) who later received second-line chemotherapy. The TAX 327 Study was the key
registration study that showed a survival benefit for docetaxel over mitoxantrone for
first-line chemotherapy treatment of patients with metastatic CRPC); |
| |
|
|
increased frequency and duration of pain palliation when adding OGX-011 to either
mitoxantrone or docetaxel as second-line chemotherapy compared to the frequency and
duration of pain palliation observed in the TAX 327 Study for first-line chemotherapy
alone in patients with CRPC; and |
| |
|
|
longer survival duration when adding OGX-011 to gemcitabine and a platinum-containing
chemotherapy compared to the survival duration reported in prior published results from
randomized clinical trials in NSCLC patients receiving gemcitabine and a
platinum-containing chemotherapy. |
6
Refer to the discussion below under the heading Summary of Results of OGX-011 Phase 2 Clinical
Trials for further details.
Final results of a randomized phase 2 trial evaluating the benefit of combining OGX-011 with
first-line docetaxel chemotherapy were presented during an oral presentation at the ASCO 2009
Annual Meeting. Analyses indicating a survival benefit in patients treated with OGX-011 in
combination with first-line docetaxel compared to docetaxel alone, the latter of which being the
current standard care for patients with advanced, progressive metastatic prostate cancer, are
described below under the heading Summary of Final Results of OGX-011 Phase 2 Clinical Trial in
First-Line Hormone Refractory Prostate Cancer.
At the 2008 ASCO Annual Meeting, OGX-011 phase 2 data in second-line chemotherapy treatment of CRPC
was reported. Durable pain palliation defined as pain palliation of 12 weeks or greater was
observed in this phase 2 trial evaluating patients with metastatic CRPC who progressed while
receiving, or within 6 months of completing, first-line docetaxel treatment. In this trial, 46% of
patients who were retreated with docetaxel as second-line treatment in combination with OGX-011 had
durable pain palliation. This is favorable even when compared to the 35% pain responses of 3 weeks
or greater observed in the phase 3 study which supported the registration of docetaxel as
first-line chemotherapy in patients with CRPC. The data from this clinical trial also showed
evidence that adding OGX-011 to docetaxel as second-line chemotherapy may have reversed docetaxel
resistance in some patients whose disease had progressed while being treated with first-line
docetaxel. In preliminary analyses, the mean average levels of serum clusterin during the OGX-011
treatment period were significantly lower compared to baseline levels before OGX-011 treatment and
low average serum clusterin levels were predictive of survival with low serum clusterin levels
correlating to longer survival.
Due to the pain palliation results observed in our phase 2 trial, the other prostate cancer phase 3
registration trial, which we intend to initiate in 2010, will evaluate the durable pain palliation
benefit of OGX-011 in patients treated with second-line chemotherapy. OncoGenex believes that its
pre-clinical and clinical data support the use of OGX-011 to improve the activity of chemotherapy
in both CRPC and NSCLC indications.
OGX-011 has received Fast Track designations from the U.S. Food & Drug Administration (FDA) for
the treatment of CRPC in combination with docetaxel for both first and second-line docetaxel
treatment. The FDA has agreed on the design of two phase 3 registration trials, each in CRPC, via
the Special Protocol Assessment (SPA) process. One trial design investigates overall survival as
the primary endpoint for OGX-011 in combination with first-line chemotherapy, whereas the other
trial design investigates pain palliation as the primary endpoint for OGX-011 in combination with
second-line chemotherapy.
OGX-011 has also received written, scientific advice from the European Medicines Agency (EMA) on
the Companys development plan for OGX-011 for the treatment of men with CRPC. The input received
from the Committee for Medicinal Products for Human Use (CHMP) at the EMA was in overall
agreement with OncoGenexs development plan regarding the proposed preclinical studies and both the
study designs and analyses for the phase III trials. The CHMP also agreed that the intended safety
database would enable a sufficiently qualified risk-benefit assessment for market approval.
OGX-011 has been administered to 294 patients with various types of cancer. Some of the patients
experienced various adverse events, the majority of which are associated with other treatments in
the protocol and the disease. The majority of adverse events were mild and the most common adverse
events associated with OGX-011 consisted of flu-like symptoms. Of the moderate and severe adverse
events associated with OGX-011, neutropenia, vomiting, diarrhea, and difficulty breathing (also
known as dyspnea) were the most common events, occurring in > 2% of patients.
The United States adopted name, or USAN, for the OGX-011 drug product is custirsen sodium.
7
Summary of OGX-011 Product Registration Strategy
As a result of the partnership with Teva, OGX-011 has committed funding for three phase 3 trials.
Two phase 3 clinical trials will evaluate the clinical benefit of OGX- 011 in CRPC. Both trials are
planned to initiate in 2010. The third phase 3 trial is being developed in collaboration with Teva,
and will evaluate the clinical benefit of OGX-011 in NSCLC. The Clinical Development Plan agreed to
by Teva and the Company under which the three phase 3 clinical trials will be initiated is as
follows:
| |
|
|
Evaluating a survival benefit for OGX-011 in combination with first-line docetaxel
treatment in approximately 800 men with CRPC, expected to initiate in the third quarter
of 2010. Teva will have primary responsibility of the oversight of this trial; |
| |
|
|
Evaluating a durable pain palliation benefit for OGX-011 in combination with docetaxel
as second-line chemotherapy in approximately 300 men with CRPC, expected to initiate
in the second quarter of 2010 subject to institutional review board approval and patient screening.
OncoGenex will have primary responsibility for the oversight of this trial; and |
| |
|
|
Evaluating a survival benefit for OGX-011 in combination with first-line chemotherapy
in at least 700 patients with NSCLC, expected to initiate in 2011. Teva will have primary
responsibility for the oversight of this trial. |
Summary of Results of OGX-011 Phase 2 Clinical Trials
Five phase 2 clinical trials have been conducted to evaluate the ability of OGX-011 to enhance the
effects of therapy in prostate, non-small cell lung and breast cancer. The following is a summary
of the clinical trials evaluating OGX-011 in combination with chemotherapy.
Summary of Final Results of OGX-011 Phase 2 Clinical Trial in First-Line Hormone Refractory
Prostate Cancer
Final results of a randomized phase 2 trial evaluating the benefit of combining OGX-011 with
first-line docetaxel chemotherapy in patients with CRPC were presented during an oral presentation
at the ASCO 2009 Annual Meeting. In this phase 2 trial, patients were randomized to receive either
docetaxel or OGX-011 plus docetaxel.
The trial enrolled 82 patients at 12 sites in Canada and the United States from September 2005 to
December 2006. Patients were randomized to one of two treatment arms to receive either 640 mg per
week of OGX-011 by intravenous infusion in combination with docetaxel and prednisone or docetaxel
and prednisone alone. Patients in both treatment arms received therapy until disease progression,
toxicity or the completion of ten 3-week cycles of therapy. Analyses indicated a survival benefit
in patients treated with OGX-011 in combination with docetaxel compared to docetaxel alone the
current standard of care for patients with advanced prostate cancer:
| |
|
|
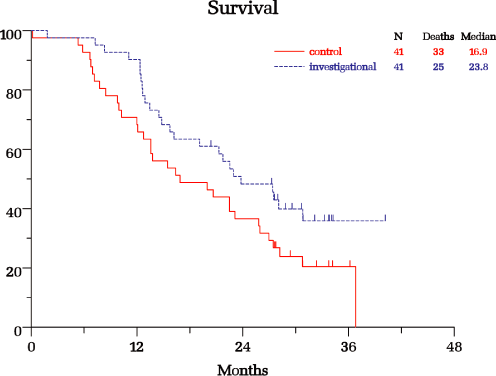
The median overall survival in patients who were treated with OGX-011 plus docetaxel
trial was 23.8 months compared to 16.9 months for patients treated with docetaxel alone,
indicating a 6.9 month survival advantage in the OGX-011 arm; |
| |
|
|
The unadjusted hazard ratio (HR), a measure used to compare the death rates between
treatment groups, was 0.61, representing a 39% lower rate of death for patients treated
with OGX-011; and |
| |
|
|
A prospectively defined multivariate analysis indicated that the significant predictors
of overall survival were treatment arm, performance status and presence of metastases
other than in bone or lymph nodes. In the multivariate analysis, patients treated with
OGX-011 had a rate of death of 51% lower than patients treated with docetaxel alone
(HR=0.49; p=0.012). Additional exploratory analyses found that the lower rate of death was
associated with the effect of OGX-011 treatment even when varying amounts of chemotherapy
were administered (i.e. OGX-011 treatment resulted
in a lower rate of death when compared to the control arm for patients receiving 6 or less
cycles of chemotherapy as well as for patients receiving 10 cycles of chemotherapy). |
8
OGX-011 treatment was well tolerated in combination with docetaxel. There was an increase in
incidence of mild fever, chills and creatinine levels (a laboratory measure for reduced kidney
function) and a moderate to significant decrease in circulating lymphocytes in the blood (another
laboratory measure) without any increase in infection rate compared to the docetaxel arm.
Lymphocytes are a type of white blood cell involved in the bodys defence against infections. Due
to the final results of this randomized phase 2 trial, the phase 3 registration trial will evaluate
the overall survival benefit of OGX-011 in patients treated with first-line chemotherapy.
The following graph display the Kaplan-Meier curves for OGX-011 in combination with docetaxel as
compared to docetaxel alone:
Summary of Results of OGX-011 Phase 2 Clinical Trial in Second-Line CRPC
We have completed accrual and patient treatment in our randomized phase 2 clinical trial in
patients with CRPC evaluating OGX-011 in combination with either docetaxel or mitoxantrone as
second-line chemotherapy. In this phase 2 trial, patients who were previously treated with a
first-line, docetaxel-based chemotherapy regimen and progressed on or within 6 months of
discontinuation of docetaxel treatment were randomized to receive OGX-011 plus either docetaxel
retreatment or mitoxantrone. Initially, 42 patients were randomized and received at least one cycle
of OGX-011 and chemotherapy and were included for analysis: 20 patients received docetaxel
retreatment plus OGX-011 and 22 patients received mitoxantrone plus OGX-011. The protocol was
amended to allow additional patients to be enrolled in the docetaxel retreatment arm. Enrollment
into the amended protocol was initiated in May 2007 and 25 additional patients were enrolled as of
September 2007. All patients received at least one cycle of OGX-011 and docetaxel retreatment and
were included in the analysis. As of June 9, 2009, the median follow-up
time for the 42 randomized patients (20 in the custirsen plus docetaxel retreatment arm and 22 in
the custirsen plus mitoxantrone treatment arm) was 30 months (range 25 to 34 months).
9
The preliminary results for the clinical trial are summarized as follows:
| |
|
|
survival duration is longer than the survival duration observed in the follow-up
evaluation of patients on the TAX 327 Study who later received second-line chemotherapy
and were available for long-term follow-up. In the follow-up evaluation of TAX 327, 237
patients received either docetaxel or mitoxantrone as second-line chemotherapy. The median
(50%) survival duration from the start of second-line chemotherapy was 10 months for both
groups of patients (patients receiving mitoxantrone as second-line chemotherapy after
receiving docetaxel as first-line chemotherapy or patients receiving docetaxel as
second-line chemotherapy after receiving mitoxantrone as first-line chemotherapy). As of
June 9, 2009, the estimated median over survival duration for the custirsen plus
mitoxantrone arm was 11.4 months (95% C.I.: 6.5-15.2 months) based on a median follow-up
of 30 months. For the custirsen plus docetaxel retreatment arm, the median overall
survival is estimated at 15.8 months (95% C.I.:9.9-23.3 months) for the 20 randomized
patients, based on a median follow-up of 30 months, and 12.8 months (95% C.I.:9.9-17.0
months) for the combined 45 patients, based on a median follow-up of 23 months; |
| |
|
|
the survival data from this clinical trial also compares favorably to the median
survival duration of 9.6 months for patients who received second-line docetaxel after
first-line docetaxel in the retrospective BCCA Study. The patients in the BCCA Study had a
better prognosis than the patients in OncoGenex clinical trial; |
| |
|
|
preliminary analyses have shown that treatment with OGX-011 in combination with
chemotherapy significantly lowers serum clusterin levels and that average serum clusterin
levels were predictive of survival with low serum clusterin levels correlating to longer
survival; and |
| |
|
|
durable pain responses (defined as a duration of 12 weeks or greater) were observed in
46% of evaluable patients in the docetaxel retreatment plus OGX-011 arm and in 46% of
patients in the mitoxantrone arm. |
Summary of Preliminary Results of OGX-011 Phase 2 Clinical Trial in NSCLC
We have completed accrual and patient treatment in our clinical trial in patients with advanced
NSCLC, evaluating OGX-011 in combination with gemcitabine and a platinum chemotherapy (cisplatin or
carboplatin) as first-line chemotherapy. In this phase 2 trial, 81 patients with advanced NSCLC
received OGX-011 in combination with gemcitabine and a platinum chemotherapy as first-line
chemotherapy. Eighty two percent of the patients had stage IV disease at enrollment. Patients are
currently being followed for survival. The preliminary results are summarized as follows:
| |
|
|
the median overall survival was 14.1 months and 54% of patients survived at least 1
year; |
| |
|
|
in January 2009, we reported that at two years, 30% of patients who had received
OGX-011 with first-line chemotherapy were alive; |
| |
|
|
for comparison, published studies using a platinum-based regimen plus gemcitabine as
first-line chemotherapy for advanced NSCLC reported median survivals of 8 to 11 months and
one-year survival rates of 33% to 43%. Market approval for Avastin plus paclitaxel and
carboplatin chemotherapy for NSCLC was based on results showing a median survival of 12.3
months compared to 10.3 months for patients treated with chemotherapy alone. Survival
rates for Avastin plus chemotherapy versus chemotherapy alone were reported as 51% versus
44% at one year and 23% versus 15% at two years, respectively; |
| |
|
|
73% of patients achieved disease control; and |
| |
|
|
preliminary analyses have shown that treatment with OGX-011 in combination with
gemcitabine and a platinum chemotherapy significantly lowers serum clusterin levels and
that average serum clusterin levels were predictive of survival with low serum clusterin
levels correlating to longer survival. |
10
Summary of Preliminary Results of OGX-011 Phase 2 Clinical Trial in Advanced Breast Cancer
In January 2009, the results of this clinical trial in patients with advanced breast cancer
evaluating OGX-011 in combination with docetaxel as first-line or second-line chemotherapy were
published in the scientific journal, Clinical Cancer Research. The authors conclusion was that the
combination of OGX-011 and docetaxel at 75 mg/m2 is well tolerated and clinical activity was seen
in these patients with metastatic breast cancer.
OGX-427
The development program for our second product candidate, OGX-427, is focused on reducing heat
shock protein 27 production to enhance treatment sensitivity and delay tumor progression in
patients who have not fully developed treatment resistance and to restore treatment sensitivity in
patients who have developed treatment resistance. Heat shock protein 27 is a protein that is
over-produced in response to many cancer treatments and which we believe promotes cell survival
based on preclinical data.
A number of pre-clinical studies conducted by the Vancouver Prostate Centre and others have shown that
inhibiting the production of Hsp27 in human prostate, breast, ovarian, pancreatic and bladder tumor
cells sensitizes the cells to chemotherapy. Pre-clinical studies conducted by The Vancouver Prostate Centre and others have shown that reducing
Hsp27 production induced tumor cell death in prostate, breast,
non-small cell lung, bladder and pancreatic cancers. The Vancouver Prostate Centre has also conducted
pre-clinical studies that indicate that reducing Hsp27 production sensitizes prostate tumor cells
to hormone ablation therapy.
A phase 1 trial has evaluated 53 patients with a variety of cancers, with enrollment ongoing.
OGX-427 was first evaluated as a single agent in a dose escalation clinical trial with doses
ranging from 200mg up to 1000mg OGX-427. A maximum tolerated dose was not identified up to and
including the 1000mg dose of OGX-427 monotherapy. Subsequently, as defined by the protocol, OGX-427
was evaluated in combination with chemotherapy in a dose escalation trial design. An 800mg dose of
OGX-427 in combination with docetaxel was evaluated, followed by a 1000mg dose of OGX-427 plus
docetaxel. A maximum tolerated dose was not identified up to and including the 1000mg dose of
OGX-427 plus docetaxel.
OGX-427 is administered as three loading doses within the first nine days and then continued
weekly, with three weeks defined as a treatment cycle, until disease progression or toxicity. In
those groups receiving OGX-427 in combination with docetaxel, 75mg/M2 docetaxel was administered on
day 1 of every 3-week cycle starting after completion of the OGX-427 loading doses.
Preliminary results of this phase 1 trial were presented during an oral presentation at the ASCO
2009 Annual Meeting. At that time, 41 patients were enrolled who had a diagnosis of breast,
ovarian, prostate or non-small cell lung cancer and most had failed multiple prior chemotherapy
treatments. A median of two cycles (range of one to eight cycles) was administered.
OGX-427 treatment was well tolerated as a monotherapy. No evidence of altered cardiac activity was
observed. A majority of adverse events were mild and mainly occurred during the loading doses.
Adverse events consisted of chills, itching and fatigue in over one-third of patients. There was a
trend for increasing incidence of some mild adverse events with escalating OGX-427 doses. For
example, 33% of patients at the 200mg dose compared to 67% of patients at the 1000mg dose had mild
adverse events during the loading doses. The half-life of OGX-427 in the blood remained constant,
although there appeared to be an increase in maximum blood levels and a corresponding decease in
blood clearance of OGX-427 as doses were escalated.
11
The combination of OGX-427 with docetaxel at both dose levels was also well tolerated. This data is
subject to further analysis.
Circulating tumor cells (CTCs), an emerging metric to assess treatment effect, were evaluated at
baseline before treatment and during OGX-427 treatment as a monotherapy. Both total and
Hsp27-positive CTCs were evaluated. Declines of 50% or greater in both total and Hsp27-positive
CTCs were observed in over one-half of the patients in each cohort and in each type of cancer.
Declines in Hsp27 CTCs to 5 or less cells occurred in 27% of patients who had greater than 5 CTCs
at baseline. Reduction in tumor markers defined as declines of prostate specific antigen, or PSA,
levels in prostate cancer or CA-125 levels in ovarian cancer were also observed. A reduction in PSA
level was observed in 7 of 20 patients (35%) with prostate cancer and a reduction in CA-125 levels
was observed in 3 of 5 patients (60%) with ovarian cancer.
An investigator-sponsored phase 1 clinical trial evaluating OGX-427 when administered directly into
the bladder in patients with bladder cancer was initiated in August 2009. The study, which will
enroll up to 36 patients with bladder cancer, is designed to determine the safety and potential
benefit of OGX-427 administered directly into the bladder using a catheter, which is known as
intravesical instillation. In addition, the study will measure the direct effect of OGX-427 on
expression of Hsp27 in bladder tumor cells as well as determine the pharmacokinetics and
pharmacodynamics of OGX-427 when delivered by intravesical instillation. This
investigator-sponsored study is funded by the National Cancer Institute of Canada (NCIC).
In January 2010, we announced that an investigator-sponsored phase 2 clinical trial evaluating
OGX-427 when administered as a monotherapy to patients with CRPC has received grant funding. The
randomized, controlled phase 2 study will enroll up to 72 patients and is designed to determine the
potential benefit of OGX-427 by evaluating the number of patients who are without disease
progression at 12 weeks post study treatment with or without OGX-427. This phase 2 trial will also
measure the direct effect of OGX-427 on PSA levels, time to progression by PSA or measurable
disease, numbers of CTCs and other relevant secondary endpoints. The trial is expected to start by
mid 2010 following final analysis of phase 1 data and approval by Health Canada and the
institutional review board. The funds were awarded by a third party granting agency to Dr. Kim Chi,
a medical oncologist at the BC Cancer Agency, Research Scientist at the Vancouver Prostate Centre and the
principal investigator of the OGX-427 phase 2 trial.
SN2310
Product candidate SN2310 is a novel camptothecin for the treatment of cancer. Camptothecins are
potent anticancer agents that belong to the family of drugs called topoisomerase I inhibitors that
bind reversibly to the TOPO-I-DNA complex causing breaks in the DNA strands during replication
resulting in cell death. The phase 1 clinical trial evaluated safety in patients with advanced
cancer who have received on average 3 to 5 prior chemotherapy treatments. The phase 1 clinical
trial has been completed and the dose-limiting toxicity that defined a maximum tolerated dose in
this heavily pretreated patient population, as expected, was a significant decrease in the number
of neutrophils (neutropenia), a type of white blood cell that is involved in the bodys defense
against infections.
SN2310 has been administered to 26 patients with various types of cancer in a phase 1 clinical
trial. Enrollment for this clinical trial has been completed. Some of the patients experienced
adverse events, which were considered unrelated to the trial drug and attributed to the underlying
disease. Of the adverse events associated with the administration of SN2310, most were mild and the
most common events were nausea, diarrhea, vomiting and fatigue. Mild to moderate reactions
(back/chest pain, flushing) have been observed during infusions. Significant neutropenia has
occurred in some patients and was the dose-limiting toxicity observed, sometimes associated with
fever or septicemia. We have not defined additional development activities for this product
candidate at this time and are currently exploring options to out-license this product candidate.
12
OGX-225
The development program for our fourth product candidate, OGX-225, is focused on reducing the
production of both IGFBP-2 and IGFBP-5 with a single product to enhance treatment sensitivity and
delay tumor progression. Increased IGFBP-2 or IGFBP-5 production is observed in many human cancers.
Increased IGFBP-2 or IGFBP-5 production is linked to faster rates of cancer progression, treatment
resistance and shorter survival duration. We believe employing OGX-225 as a single product to
simultaneously inhibit the production of both IGFBP-2 and IGFBP-5 has the potential to delay
disease progression in cancers dependent upon insulin-like growth factor 1 (IGF-1), for
proliferation. OncoGenex has completed pre-clinical proof of concept studies with OGX-225.
We believe that because IGFBP-2 and IGFBP-5 are over-produced in a variety of cancers, OGX-225 may
have broad market potential to treat many cancer indications. OncoGenex believes that the initial
opportunity for OGX-225 would be in breast and prostate cancer patients early in the course of
their recurrence after failed hormone ablation therapy.
We have identified the lead compound and numerous pre-clinical proof of concept studies with
OGX-225 have been completed indicating that it delays progression to hormone independence in
prostate and breast cancer model systems. We have not defined when it will initiate the
pre-clinical studies required for a regulatory submission and initiation of phase 1 clinical
trials.
CSP-9222
Product candidate CSP-9222 is the lead compound from a family of caspase activators. These novel,
small molecules have been identified as activators of programmed cell death in pre-clinical models.
Summary of Product Development Programs
The following table summarizes the status of our product development programs:
| |
|
|
|
|
|
|
|
|
| |
|
Cancer Indication and |
|
|
|
|
|
Recent and Expected Near Term Data |
| Product Candidate |
|
Study |
|
Treatment Combination(1) |
|
Development Phase/Status |
|
Releases |
OGX-011 phase 3 (2)
|
|
Castrate Resistant Prostate Cancer Survival Endpoint
(OGX-011-11)
|
|
First-line docetaxel with and without OGX-011 (~800 patients)
|
|
Initiation pending; SPA approved by the FDA
and EMA in agreement with development plan
|
|
Expected to initiate in the third quarter of 2010. Teva has primary responsibility
for the oversight of this trial |
|
|
Castrate Resistant Prostate Cancer Durable Pain
Palliation Endpoint
(OGX-011-10)
|
|
Docetaxel as second-line chemotherapy with and without OGX-011(~300
patients)
|
|
Initiation pending; SPA approved by the FDA
and EMA in agreement with development plan
|
|
Expected to initiate in the second quarter of 2010. OncoGenex has primary
responsibility for the oversight of this trial |
|
|
Advanced Non-Small Cell Lung Cancer Survival
Endpoint
|
|
Chemotherapy with or without OGX-011 (~ 700 patients)
(Protocol to be determined)
|
|
Initiation pending
|
|
Expected to initiate in early 2011. Teva has primary
responsibility for the oversight of this trial |
OGX-011 phase 2
|
|
Castrate Resistant Prostate Cancer
|
|
First-line docetaxel with and without OGX-011
|
|
Phase 2 completed
|
|
Final survival data presented at 2009 ASCO Annual Meeting |
|
|
(OGX-011-03)
|
|
|
|
|
|
Manuscript submitted |
|
|
Castrate Resistant Prostate Cancer
(OGX-011-07)
|
|
OGX-011 with second-line chemotherapy (docetaxel or mitoxantrone)
|
|
Phase 2 completed
|
|
Interim data presented at ASCO GU Symposium 2008 Manuscript in
preparation |
|
|
|
|
|
|
|
|
Manuscript in preparation |
|
|
Castrate Resistant Prostate Cancer
(OGX-011-07A)
|
|
OGX-011 with docetaxel as second-line chemotherapy
|
|
Phase 2 completed
|
|
Manuscript in preparation |
|
|
Advanced Non-Small Cell Lung Cancer
(OGX-011-05)
|
|
OGX-011 with first-line chemotherapy (gemcitabine and cisplatin or
gemcitabine and carboplatin)
|
|
Phase 2 completed
|
|
2-year survival data presented Feb 2009
Manuscript in preparation
|
|
|
Localized Prostate Cancer
(OGX-011-04)
|
|
OGX-011 with hormone ablation therapy
|
|
Phase 2 completed
|
|
Results presented at ASCO GU 2008 |
|
|
Advanced Breast Cancer
(OGX-011-06)
|
|
OGX-011 with docetaxel as second-line chemotherapy
|
|
Phase 2 completed
|
|
Data published in Clinical Cancer Research 2009 |
OGX-011 phase 1
|
|
Localized Prostate Cancer
(OGX-011-01)
|
|
OGX-011 with hormone ablation therapy
|
|
Phase 1 completed
|
|
Data published in Journal of National Cancer Institute 2005 |
|
|
Solid Tumors
(prostate, breast, NSCL, ovarian, renal, bladder,
peritoneum)
(OGX-011-02)
|
|
OGX-011 with docetaxel
|
|
Phase 1 completed
|
|
Data published in Clinical Cancer Research 2008 |
OGX-427 phase 2
|
|
Castrate Resistant Prostate Cancer
|
|
OGX-427 as monotherapy
|
|
Not yet initiated
|
|
Expected to initiate in 2010 |
OGX-427 phase 1
|
|
Solid Tumors
|
|
OGX-427 with and without chemotherapy
|
|
Phase 1 ongoing- accrual and treatment
complete for evaluation of OGX-427 as monotherapy. On-going accrual
and treatment ongoing for evaluation of OGX-427 with chemotherapy
|
|
Monotherapy evaluation complete maximum tolerated dose not
determined at highest dose level. |
|
|
|
|
|
|
|
Data presented at 2009 ASCO Annual Meeting |
|
|
|
|
|
|
|
Complete study in 2010 and present data if selected at ASCO
2010 |
|
|
Bladder Cancer
|
|
OGX-427 as monotherapy
|
|
Phase 1 ongoing
|
|
None |
SN2310
|
|
Solid Tumors
|
|
SN2310 administered to heavily pre-treated patients with advanced cancer
|
|
Phase 1 completed
|
|
Enrollment and treatment completed maximum tolerated dose determined |
OGX-225
|
|
Solid Tumors
|
|
OGX-225 with and without chemotherapy
|
|
Pre-clinical proof-of-concept studies completed
|
|
None |
CSP-9222
|
|
Solid Tumors
|
|
To be determined
|
|
Formulation to be determined
|
|
None |
13
| |
|
|
| (1) |
|
In all of our prostate cancer clinical trials and in clinical practice for prostate cancer,
docetaxel is administered in combination with prednisone. |
| |
| (2) |
|
We have designed two phase 3 clinical trials to evaluate the clinical benefit of OGX-011 in
metastatic CRPC. OncoGenex believes that two phase 3 trials will be required for initial product
marketing approval. |
Overview of Market and Treatment
In North America, cancer is expected to strike slightly fewer than one in two men and slightly more
than one in three women in their lifetimes and has recently surpassed heart disease as the leading
cause of death in the United States. The American Cancer Society estimated that in 2009
approximately 1,479,350 new patients in the United States would be diagnosed with cancer and that
there would be approximately 562,340 patient deaths attributable to cancers.
Typically, cancer treatment is given sequentially and can include surgery, radiation therapy,
chemotherapy and hormone therapy. Although a particular therapy may initially be effective, tumor
cells often react to therapeutic treatment by increasing the production of proteins that afford
them a survival advantage, which enable them to become resistant to therapy, multiply and spread to
additional organs. As a result, many patients progress rapidly through all available therapies and
ultimately die.
Our Strategy
| |
|
|
Focus on gaining market approval for OGX-011 by conducting registration trials, in both
prostate and lung cancer, in collaboration with Teva, that demonstrate efficacy and
safety. As a result of the Collaboration Agreement with Teva, committed funding is
available for phase 3 trials in 1st line CRPC, 2nd line CRPC, and
1st line NSCLC. The prostate trials are intended to begin in 2010 and the NSCLC
trial is expected to begin in early 2011. Currently, both mitoxantrone and docetaxel are
approved for use in patients with CRPC, but docetaxel was shown to improve patient
survival when compared to mitoxantrone. Currently, Avastin, carboplatin, gemcitabine and
cisplatin are approved for use in patients with NSCLC, but Avastin in combination with
paclitaxel and carboplatin chemotheraphy was shown to improve patient survival when
compared to carboplatin chemotherapy alone. |
| |
|
|
Advance OGX-427 by conducting clinical trials across multiple cancer indications for
OGX-427 including, but not limited to, bladder cancer and CRPC. Consistent with the
strategy OncoGenex is following for OGX-011, OncoGenex intends to conduct parallel
clinical trials to evaluate OGX-427 in several cancer indications and treatment
combinations to accelerate its assessment of this product candidate for further
development. |
14
| |
|
|
Focus on developing and commercializing new cancer therapies to inhibit treatment
resistance in cancer patients. OncoGenex plans to leverage its expertise in discovery and
development to bring new products to market as fast as possible. OncoGenex intends to
maintain and develop its relationships with the Vancouver Prostate Centre, and develop relationships
with other research institutions in order to identify and source additional product
candidates. |
| |
|
|
Optimize the development of our product candidates through use of outsourcing and
internal expertise. In order to increase efficiency and lower its overhead, OncoGenex
outsources, and plans to continue to outsource, pre-clinical and manufacturing activities.
OncoGenex has chosen to establish critical product development functions in-house,
including clinical trial management and regulatory affairs. |
Second Generation Antisense Technology
OGX-011, OGX-427 and OGX-225 are based on second-generation antisense drug chemistry and belong to
the drug class known as antisense therapeutics. On a product by product basis, OncoGenex has
collaborated with Isis Pharmaceuticals Inc. (Isis) and selectively licensed technology from Isis
to combine Isis second generation antisense chemistry with our proprietary gene target sequences
to create inhibitors which are designed to down regulate certain proteins associated with cancer
resistance. In contrast to first-generation antisense chemistry, second-generation antisense
chemistry has improved target binding affinity, increased resistance to degradation and improved
tissue distribution. These improvements result in slower clearance of the therapies from the body,
allowing for less frequent dosing and thereby making treatment easier on patients at a lower
associated cost. For example, clinical data from our phase 1 clinical trial in prostate cancer
patients demonstrated that weekly intravenous administration of OGX-011 resulted in drug
distribution to prostate cancer tissue and over 92% inhibition of its target, clusterin mRNA, in
prostate tumor cells in these patients. These data demonstrate that following systemic
administration, OGX-011 entered tumor cells and inhibited clusterin production.
License and Collaboration Agreements
Teva Pharmaceutical Industries Ltd.
OGX-011
As discussed above, in December 2009, OncoGenex, through its wholly-owned subsidiary, OncoGenex
Technologies, entered into the Collaboration Agreement with Teva for the development and global
commercialization of OGX-011 (and related compounds). Pursuant to the Collaboration Agreement, Teva
received the exclusive worldwide right and license to develop and commercialize any products
containing OGX-011 and related compounds (Licensed Products). We have an option to co-promote
OGX-011 in the United States and Canada.
Under the Collaboration Agreement, Teva made upfront payments in the aggregate amount of $50
million, will pay up to $370 million upon the achievement of developmental and commercial
milestones and will pay royalties at percentage rates ranging from the mid-teens to mid-twenties on
net sales, depending on aggregate annual net sales of Licensed Products. In addition to the ongoing
royalties, Teva will pay us additional one-time sales threshold royalties if certain aggregate net
sales are achieved. We are required to contribute $30 million towards the development of OGX-011
which will include personnel costs for certain development activities.
15
Teva and OncoGenex have developed a Clinical Development Plan under which the following three phase
3 clinical trials will be initiated:
| |
|
|
a phase 3 clinical trial of OGX-011 in approximately 300 patients with second-line
castrate resistant prostate cancer, expected to initiate in the second quarter of 2010 subject to institutional review board approval and patient screening. OncoGenex will have
primary responsibility for the oversight of this trial; |
| |
|
|
a phase 3 clinical trial of OGX-011 in approximately 800 patients with first-line
castrate resistant prostate cancer, expected to initiate in the third quarter of 2010. Teva will have primary
responsibility for the oversight of this trial; and |
| |
|
|
a phase 3 clinical trial of OGX-011 in at least 700 patients with first-line NSCLC,
expected to initiate by early 2011. Teva will have primary responsibility for the
oversight of this trial. |
Teva will be responsible for conducting any other studies and development work necessary to obtain
required regulatory approvals. We may assume some of these activities if assigned by the Joint
Steering Committee, but Teva would be responsible for all associated costs. The Joint Steering
Committee will oversee the development and regulatory approval of any Licensed Product. Funding
responsibilities for the Clinical Development Plan will be allocated as follows:
| |
|
|
We will be required to contribute $30 million towards the development of OGX-011 which
will include personnel costs for certain development activities; and |
| |
|
|
Teva will be required to fund all other expenses under the Clinical Development Plan. |
In addition to the development costs noted above, Teva is also responsible for all costs relating
to product commercialization including any costs incurred in relation to our co-promotion option,
except for start-up costs in advance of commercialization.
Teva was also granted the first right to file, prosecute and maintain, and enforce at its expense
the patent rights associated with OGX-011. If Teva elects, however, not to or fails to file,
prosecute and maintain, and enforce the patent rights associated with OGX-011, OncoGenex retains
the right to assume responsibility for the filing, prosecution and maintenance, and enforcement of
the patent rights associated with OGX-011.
The Collaboration Agreement will remain in effect, on a country-by-country basis, until the
expiration of the obligation of Teva to pay royalties on sales of the Licensed Product in such
country (or earlier termination under its terms). Commencing after the completion of all three
phase 3 clinical trials set forth in the Clinical Development Plan, or upon early termination due
to a material adverse change in our patent rights related to OGX-011 or safety issues or futility
as defined in the Collaboration Agreement, Teva may terminate the Collaboration Agreement in its
sole discretion upon three months notice if notice is given prior to regulatory approval of a
Licensed Product and upon six months notice if notice is given after such regulatory approval. If
Teva terminates the Collaboration for any reasons other than an adverse change in OGX-011 patent
rights, safety issues or futility determination as previously described, it will remain
responsible for paying for any remaining costs of all three phase 3 clinical trials, except for
Company Development Expenses. Either party may terminate the Collaboration Agreement for an uncured
material breach by the other party or upon the bankruptcy of either party. If the Collaboration
Agreement is terminated other than for an uncured material breach by Teva, we will pay Teva a
royalty on sales of Licensed Products. The percentage rates of such royalties (which are in the
single digits) depend if termination occurs prior to the first regulatory approval in the United
States or a primary European Market or after one of these approvals. These royalties would expire
on a country-by-country basis on the earlier of ten years after the first commercial sale of a
Licensed Product or certain thresholds related to generic competition.
In the event of a change of control of the Company and within 90 days of the change of control,
Teva may terminate the Joint Steering Committee in its sole discretion, terminate the co-promotion
option in its sole discretion if not then exercised by us or if exercised but not yet executed by
us, or terminate the co-promotion option if in its commercially reasonable opinion co-promotion
with our successor would be materially detrimental to Tevas interests.
16
Isis Pharmaceuticals, Inc.
OGX-011
In November 2001, OncoGenex Technologies entered into an agreement with Isis to jointly develop and
commercialize OGX-011 (Original Isis Agreement). This strategic relationship provided us with
access to Isis proprietary position in second-generation antisense chemistry for use in OGX-011,
Isis expertise in developing antisense therapeutics, including their manufacturing expertise, and
allowed us to develop OGX-011 cost efficiently. Under the Original Isis Agreement, we shared with
Isis, on a basis of 65% OncoGenex and 35% Isis, the costs and revenues resulting from the
development and commercialization of OGX-011. On July 2, 2008, OncoGenex and Isis amended the
Original Isis Agreement (Amended Isis Agreement) pursuant to which OncoGenex became solely
responsible for the costs and development of OGX-011, and, in turn, took on certain financial
obligations to Isis, primarily related to sharing revenues received by OncoGenex from a third party
as a result of a licensing transaction.
Licensing revenues that are based on a percentage of net sales of a licensor are defined as Royalty
Revenue, while other licensing revenues, with the exception of fair market value of equity and
reimbursement of research and development expenses, are defined as Non-Royalty Revenue. We will pay
Isis royalties comprised of a base percentage of net sales of OGX-011 and a percentage of Royalty
Revenue we receive in excess of a certain threshold up to a certain cap. The amount of the
royalties payable to Isis is dependent on whether Isis owes royalty payments to third parties
pursuant to its license agreements with third parties. Based on the Royalty Revenue we are eligible
to receive as a result of the royalty rates established in our collaboration with Teva, our total
royalty obligations to Isis will range between 6.38% and 7.00% of net sales of OGX-011 by Teva
during the period Isis owes royalty payments to third parties and between 3.88% and 4.50% when
those third party obligations of Isis have expired, which we expect to occur in 2017.
OncoGenex paid Isis $10 million in January 2010 as Isis share of Non-Royalty Revenue received by
OncoGenex in December 2009 in connection with our Collaboration Agreement with Teva. OncoGenex
does not anticipate making any further payments to Isis in 2010 under the terms of the Amended Isis
Agreement.
In addition, we are required to pay to Isis 30% of all Non-Royalty Revenue we receive. Isis has
disclosed in its SEC filings that it is entitled to receive 30% of the up to $370 million in
milestone payments we may receive from Teva as part of the Collaboration Agreement; however, we
believe that certain of the milestone payments related to sales targets may qualify as Royalty
Revenue, and therefore be subject to the lesser payment obligations as discussed above. No
assurance can be provided that we will be entitled to receive these milestone payments or, if we
are, that the applicable amount payable to Isis will be less than 30%. Neither OncoGenex nor Isis
can pursue the development or commercialization of any antisense compound for clusterin outside of
the Amended Isis Agreement. This arrangement will continue until OGX-011 is no longer being
developed or commercialized or until the agreement is terminated early due to an uncured material
breach.
To facilitate the execution and performance of the Collaboration Agreement, OncoGenex and Isis
agreed to amend the Amended Isis Agreement (the Second Isis Amendment). The Second Isis
Amendment provides that, among other things, if the Company is the subject of a change of control
with a third party, where the surviving company immediately following such change of control has
the right to develop and sell the product, then (i) a milestone payment of $20 million will be due
and payable to Isis 21 days following the first commercial sale of the product in the United
States; and (ii) unless such surviving entity had previously sublicensed the product and a royalty
rate payable to Isis by the Company has been established, the applicable royalty rate payable to
Isis will thereafter be the maximum amount payable under the Amended Isis Agreement. Any
non-royalty milestone amounts previously paid will be credited toward the $20 million milestone if
not already paid. As a result of the $10 million milestone payment paid to Isis in relation to the
Collaboration Agreement, the remaining amount owing in the event of change of control discussed
above is a maximum of $10 million. As the Company has now licensed the product to Teva and
established a royalty rate payable to Isis, no royalty rate adjustments would apply if Teva
acquires the Company and is the surviving company.
17
OncoGenex Technologies has agreed to indemnify Isis and persons affiliated with Isis against
liabilities resulting from the development, manufacture, use, handling, storage, sale or other
commercialization or disposition of OGX-011, caused by OncoGenex Technologies or its licensees or
sublicensees gross negligence or willful misconduct, or caused by OncoGenex Technologies material
breach of the agreement.
OGX-427
In January 2005, OncoGenex Technologies entered into a collaboration and license agreement with
Isis to jointly identify antisense compounds designed to inhibit the production of proteins encoded
by specified gene targets. OncoGenex Technologies is solely responsible for all product development
activities for antisense compounds under this collaboration. This relationship provides OncoGenex
Technologies with access to Isis proprietary position in second generation antisense chemistry for
use in specified products. OncoGenex Technologies was permitted to designate up to two
collaboration gene targets for collaborative research, development and commercialization. In April,
2005, Hsp27 was confirmed as a collaboration gene target. OncoGenex and Isis jointly designed and
screened antisense compounds for this gene target. OncoGenex Technologies right to designate a
second collaboration gene target expired on January 5, 2007.
Under the terms of the agreement, in the event that OncoGenex Technologies abandons OGX-427, Isis
may elect to unilaterally continue development of OGX-427, in which case it must provide Isis with
a worldwide license or sublicense (as the case may be) of its relevant technology solely to develop
and commercialize OGX-427 in exchange for a royalty on Isis sales of OGX-427.
In consideration for the grant of rights related to OGX-427, on May 5, 2005 OncoGenex Technologies
issued Isis a promissory note which was converted into shares of OncoGenex Technologies that were
exchanged in the Arrangement for 53,200 OncoGenex Pharmaceuticals common shares. Under the terms of
the agreement, OncoGenex Technologies may be obligated to make certain milestone payments to Isis
contingent upon the occurrence of certain clinical development and regulatory events related to
OGX-427. It is also obligated to pay to Isis certain milestone payments as well as certain
royalties on net sales for OGX-427, with the amount of royalties depending on whether third party
royalty payments are owed. We
anticipate paying Isis $750,000 in 2010 upon the initiation of a phase 2 clinical trial of OGX-427
in patients with CRPC. We do not anticipate making any royalty payments to Isis under the terms of
the agreement in 2010.
OncoGenex Technologies has agreed to indemnify Isis and certain persons affiliated with Isis
against liabilities caused by its and its licensees and sublicensees gross negligence or willful
misconduct, its material breach of the collaboration and license agreement, and the manufacture,
use, handling, storage, sale or other disposition of OGX-427 that is sold by OncoGenex Technologies
or its affiliates, agents or sublicensees.
The term of the agreement will continue for each product until the later of 10 years after the date
of the first commercial sale of OGX-427, or the expiration of the last to expire of any patents
required to be licensed in order to use or sell OGX-427, unless OncoGenex Technologies abandons
OGX-427 and Isis does not elect to unilaterally continue development of OGX-427.
University of British Columbia
OGX-011
Under an agreement made in November 2001, as amended, the University of British Columbia (UBC)
granted to OncoGenex Technologies an exclusive, worldwide license to commercialize its existing
intellectual property and any improvements related to clusterin. This technology combined with
Isis second-generation antisense chemistry is our product candidate, OGX-011. In connection with
entering into this license agreement, we issued to UBC shares of OncoGenex Technologies that were
exchanged in the Arrangement for 15,243 common shares of OncoGenex Pharmaceuticals. OncoGenex
Technologies agreed to pay to UBC certain royalties on milestones and the revenue from sales of
OGX-011. OncoGenex Technologies is obligated to pay to UBC CAD$2,000 in annual maintenance fees. In
January 2010, we paid UBC $333,333 as a result of upfront payments we received from Teva in
December 2009 in connection with our Collaboration Agreement. The occurrence and receipt of future
milestone payments and the generation of royalty revenue are uncertain.
18
OncoGenex Technologies agreed to use its commercially reasonable efforts to develop and exploit the
licensed technology and any improvements. OncoGenex Technologies also agreed to promote, market and
sell any resulting products and to cause the market demand for such products to be satisfied.
OncoGenex Technologies is permitted to sublicense the technology, subject to certain consent and
other requirements. OncoGenex Technologies directs patent prosecution and is responsible for all
fees and costs related to the preparation, filing, prosecution and maintenance of the patent rights
underlying the agreement. OncoGenex Technologies indemnifies UBC and certain of UBCs affiliates
against liability arising out of the exercise of any rights granted pursuant to the agreement. The
term of this agreement will expire on the later of 20 years from its effective date or the expiry
of the last patent licensed under the agreement. Subject to patent term extensions, the current
granted patent for OGX-011 expires in the United States in 2021 and would expire in all other
jurisdictions by 2020. OncoGenex Technologies has additional patent applications pending which, if
issued and not invalidated, may extend the expiration date of the last-to-expire patents. OncoGenex
Technologies may also file additional patent applications related to clusterin that could
potentially extend the expiration date of the last to expire patent in this area.
OGX-427
Under an agreement made in April 2005, as amended, UBC granted to OncoGenex Technologies an
exclusive, worldwide license to commercialize its existing intellectual property and any
improvements related to Hsp27. This technology combined with Isis second-generation antisense
chemistry is our product candidate, OGX-427. In connection with entering into this license
agreement, OncoGenex Technologies issued to UBC shares that were exchanged in the Arrangement for
6,533 common shares of OncoGenex Pharmaceuticals. OncoGenex Technologies also agreed to pay to UBC
certain royalties on the revenue from sales of OGX-427, which royalty rate may be reduced in the
event that OncoGenex Technologies must pay additional royalties under patent licenses entered into
with third parties in order to
manufacture, use or sell OGX-427. OncoGenex Technologies may be obligated to make milestone
payments to UBC contingent upon the occurrence of certain clinical development and regulatory
events related to OGX-427. OncoGenex Technologies is obligated to pay to UBC CAD$2,000 in annual
maintenance fees. We anticipate paying UBC CAD$100,000 in 2010 upon the initiation of a phase 2
trial of OGX-427 in patients with CRPC. The occurrence and receipt of upfront and milestone
payments and the generation of royalty revenue are uncertain.
Subject to certain exceptions, OncoGenex Technologies agreed to use its commercially reasonable
efforts to (i) develop and exploit the licensed technology and any improvements, and (ii) promote,
market and sell any resulting products. OncoGenex Technologies is permitted to sublicense the
technology, subject to certain consent and other requirements. OncoGenex Technologies directs
patent prosecution and is responsible for all fees and costs related to the preparation, filing,
prosecution and maintenance of the patent rights underlying the agreement. OncoGenex Technologies
indemnifies UBC and certain of UBCs affiliates against liability arising out of the exercise of
any rights granted pursuant to the agreement. The term of this agreement will expire on the later
of 20 years from its effective date or the expiry of the last patent licensed under the agreement.
Depending on the outcome of the pending patent applications in the licensed patent family, and
subject to any applicable patent term extensions, a patent issuing from this family would expire in
all jurisdictions by 2023. OncoGenex Technologies may also file additional patent applications
related to Hsp27 that could potentially extend the expiration date of the last to expire patent in
this area.
19
OGX-225 and CSP-9222
Pursuant to the terms of our third party license agreements relating to OGX-225 and CSP-9222,
OncoGenex will owe payments upon the completion of product development milestones as well as
royalties on product sales. We do not anticipate making any milestone or royalty payments to third
parties under the terms of these agreements in 2010.
OncoGenex is also obligated to pay annual license fees to third parties with respect to these
product candidates. These amounts are disclosed in Managements Discussion and Analysis of
Financial Condition and Results of Operations Contractual Obligations, which is incorporated
herein by reference.
Summary of Milestone Obligations by Product Candidate
The following table sets forth the milestones that we may be required to pay to third parties under
the license and collaboration agreements described above. As described above, we will also
required to pay certain revenue-based royalties with respect to each of our product candidates.
| |
|
|
|
|
| Milestone Obligations to Third Parties |
|
Amount Payable |
| |
OGX-011 |
|
31% of non-royalty revenue |
OGX-427 |
|
Up to $5,813,000 (1)(2)(3) |
OGX-225(3) |
|
Up to $4,313,000 (2)(3) |
CSP-9222 |
|
Up to $14,000,000 (4) |
| |
|
|
| (1) |
|
Additional milestone payments may be required in respect of OGX-427 for product approvals
outside the field of oncology. |
| |
| (2) |
|
Payable in connection with the initiation of certain clinical trials and obtaining certain
market approvals. |
| |
| (3) |
|
Certain milestone payments are payable in Canadian dollars, which are translated based on the
December 31, 2009 exchange rate of US$1.00 = CAD$1.047, and rounded to the nearest $1,000. |
| |
| (4) |
|
Payable in connection with the initiation of certain clinical trials, making certain regulatory
filings and obtaining certain market approvals. |
Government RegulationsDrug Approval Process
Regulation by governmental authorities in the U.S. and other countries is a significant factor in
our ongoing research and development activities and in the production and marketing of our
products. In order to undertake clinical trials, to produce and market products for human use,
mandatory procedures and safety standards, established by the FDA in the U.S. and by comparable
agencies in other countries, must be followed.
The standard process before a pharmaceutical agent may be marketed includes the following steps:
| |
|
|
preclinical studies including laboratory evaluation and animal studies to test for
initial safety and efficacy; |
| |
|
|
submission to national health authorities of an Investigational New Drug (IND) IND,
or Clinical Trials Application (CTA) or equivalent dossier, which must be accepted by
each national health authority before human clinical trials may commence in that country; |
| |
|
|
adequate and well-controlled clinical trials to establish the safety and efficacy of
the drug in its intended population and use(s); |
| |
|
|
submission to appropriate national and/or regional regulatory health authorities of a
NDA, or equivalent marketing authorization application, which application is not
automatically accepted for review; and |
| |
|
|
approval by appropriate regulatory health authorities of the marketing authorization
application prior to any commercial sale or shipment of the drug in each country or
jurisdiction. |
20
As part of the regulatory health authority approval for each product, the drug-manufacturing
establishment is subject to inspection by the FDA and must comply with current Good Manufacturing
Practices (cGMP) requirements applicable to the production of pharmaceutical drug products. The
facilities, procedures, and operations of manufacturers must be determined to be adequate by the
FDA before product approval.
Preclinical studies include laboratory evaluation of the active drug substance and its formulation
in animal studies to assess the potential safety and efficacy of the drug and its formulation.
Prior to initiating the first clinical testing of a new drug product candidate, the results of the
preclinical studies are submitted to regulatory health authorities as part of an IND or CTA, and
must be accepted before the proposed clinical trial(s) can begin.
Clinical trials for cancer therapeutics involve the administration of the investigational drug
product to patients with a defined disease state, under the supervision of a qualified principal
investigator.
Clinical trials are conducted in accordance with protocols that detail the parameters to be used to
monitor safety and efficacy. Each protocol is submitted to regulatory health authorities as part of
the IND/CTA, in each country where clinical trials are to be conducted. Each clinical trial is
approved and monitored by independent Institutional Review Boards or Ethics Committees who consider
ethical factors, informed consent documents, the safety of human subjects and the possible
liability of the institutions conducting a clinical trial. The Institutional Review Board or Ethics
Committee may require changes in the clinical trials protocol, which may delay initiation or
completion of the trial.
Clinical trials typically are conducted in three sequential phases, although the phases may
overlap. In phase 1, the initial introduction of the drug to humans, the drug is tested for safety
and clinical pharmacology. Phase 2 trials involve more detailed evaluation of the safety and
efficacy of the drug in patients with a defined disease. Phase 3 trials consist of large scale
evaluations of safety and efficacy of the investigational product compared to accepted standard
therapy in a defined disease.
The process of completing clinical testing and obtaining regulatory health authority approval for a
new product takes a number of years and requires the expenditure of substantial resources.
Regulatory health authorities may conclude that the data submitted in a marketing authorization
application are not adequate
to support an approval and may require further clinical and preclinical testing, re-submission of
the application, and further review. Even after initial approval has been obtained, further studies
may be required to provide additional data about the approved indication, and further studies will
be required to gain approval for the use of a product for clinical indications other than those for
which the product was approved initially. Also, health authorities require post-marketing
surveillance programs to monitor the drug products side effects.
Marketing of pharmaceutical products outside of the U.S. is subject to regulatory requirements that
vary from country to country. In the European Union, the general trend has been towards
coordination of common standards for clinical testing of new drug products. Centralized approval in
the European Union is coordinated through the EMA.
The level of regulation outside the U.S. and European Union varies widely. The time required to
obtain regulatory approval from regulatory agencies in each country may be longer or shorter than
that required for FDA or EMA approval. In addition, in certain markets, reimbursement is subject to
governmentally mandated prices.
Many of the chemicals and compounds used in our research and development efforts are classified as
hazardous materials under applicable federal, state and local environmental laws and regulations.
We are subject to regulations under state and federal law regarding occupational safety, laboratory
practices, handling and disposing of chemicals, environmental protection and hazardous substance
control.
21
Contract Research Agreements
Consistent with our strategy to outsource certain product development activities, we have
established contract research agreements for pre-clinical, manufacturing and some data management
services. We choose which business or institution to use for these services based on their
expertise, capacity and reputation and the cost of the service.
We also provide quantities of our product candidates to academic research institutions to
investigate the mechanism of action and evaluate novel combinations of its product candidates with
other cancer therapies in various cancer indications. These collaborations expand our research
activities for product candidates with modest contribution from OncoGenex.
Research and Development Expenditures
For the years ended December 31, 2009, 2008 and 2007, our expenditures for research and development
activities were $24.2 million, $7.8 million, and $4.1 million, respectively. Such research and
development expenses primarily related to the advancement of our lead product candidate, OGX-011.
Manufacturing
We do not own facilities for the manufacture of materials for clinical or commercial use. We rely
and expect to continue to rely on contract manufacturers to manufacture our product candidates in
accordance with cGMP, for use in clinical trials. We will ultimately depend on contract
manufacturers for the manufacture of our products, when and if it has any, for commercial sale, as
well as for process development as required.
To date, all active pharmaceutical ingredient (API), for OGX-011 has been manufactured by Isis or
Avecia Biotechnology Inc. (Avecia) on a purchase order basis, under cGMP. Drug product
manufactured from API has been performed by Formatech, Inc., Pyramid Laboratories Inc., and
Laureate Pharma, Inc., in several separate manufacturing campaigns, pursuant to purchase orders or
short-term contracts with OncoGenex or its licensors. For OGX-427, all API has been manufactured
for OncoGenex by Avecia and all drug product has been manufactured for OncoGenex by Laureate
Pharma, Inc., in each case pursuant to a purchase order or short-term contract that has been
fulfilled. Contract manufacturing for commercial product is being evaluated and may or may not be
performed at the current manufacturers. Larger contract
manufacturers that can meet higher commercial drug quantities may be required and contracted to
manufacture our products for commercial sale, when and if it has any.
Intellectual Property
Our success depends in part on the ability of us and our collaborators to obtain and maintain
proprietary protection for our product candidates, technology and know-how; prevent others from
infringing the proprietary rights for our product candidates; and operate without infringing on the
proprietary rights of others.
For each of OGX-011, OGX-427 and OGX-225, our intellectual property results from our licenses with
UBC and Isis. In addition, Isis has assigned a three-member patent family related to clusterin
antisense to OncoGenex Technologies, and OncoGenex has a pending application on an OGX-427
formulation.
As discussed above, certain intellectual property rights relating to OGX-011 have been sublicensed
exclusively to Teva. Teva will subsequently be taking over control of the prosecution of those
rights in a graduated fashion, as OGX-011 is developed.
OncoGenex has been granted non-exclusive rights to all intellectual property owned, licensed or
otherwise controlled by Isis at the date of its agreements with Isis that relate to
second-generation antisense chemistry and that are required for its product candidates (such as
OGX-011, OGX-427 and OGX-225). Isis is generally restricted from engaging in research, development
and commercialization of antisense compounds related to clusterin, Hsp27, IGFBP-5 and IGFBP-2,
other than as provided in the collaboration and license agreement related to each target. Isis
directs patent prosecution and is responsible for all fees and costs related to the preparation,
filing, prosecution and maintenance of these patent rights, which extend to numerous jurisdictions
throughout the world. Individual patents have terms of protection depending on the laws of the
countries in which the applications are made.
22
All TOCOSOL and SN2310 intellectual property is owned by OncoGenex Pharmaceuticals, Inc, and
intellectual property relating to CSP-9222 is licensed from Bayer.
For intellectual property under license from UBC and Bayer covering OGX-427, OGX-225, and CSP-9222,
OncoGenex directs patent prosecution and is responsible for all fees and costs related to the
preparation, filing, prosecution and maintenance of the patent rights underlying the agreement. For
this intellectual property, OncoGenex files patent applications in the United States, Canada,
Europe (through the European Patent Office), Japan, and other jurisdictions.
Composition of matter patents covering OGX-011, OGX-427, SN2310, CSP-9222 and TOCOSOL have issued
in the U.S. and certain other jurisdictions. Additional patent applications covering all of these
products, as well as other technologies, are pending in the U.S. and certain other countries.
Generally, patents issued in the U.S. are effective for 20 years from the earliest non-provisional
filing date, if the application from which the patent issues was filed on or after June 8, 1995
(otherwise the term is the longer of 17 years from the issue date or 20 years from the earliest
non-provisional filing date). The duration of patent terms for non-U.S. patents is typically 20
years from the earliest corresponding national or international filing date. OncoGenex licensed
UBC patent estate, based on those patents and applications existing now and expected by OncoGenex
to issue, will expire in years ranging from 2020 to 2024, such dates do not include extensions that
may be available. Our TOCOSOL patent terms will expire starting from 2018, and the SN2310 and
CSP-9222 patent terms from 2023. Patent term extensions, specifically to make up for regulatory
delays, are available in the U.S., Europe, and Japan. Although we believe that some or all of its
product candidates will meet the criteria for patent term extensions, there can be no assurance
that it will obtain such extensions.
We also rely on unpatented trade secrets, proprietary know-how and continuing technological
innovation, which we seek to protect, in part, by confidentiality agreements with our corporate
partners, collaborators, employees and consultants in our drug development research. There can be
no assurance that these
agreements will not be breached, that we will have adequate remedies for any breach, or that our
trade secrets or know-how will not otherwise become known or be independently discovered by
competitors. Further, there can be no assurance that we will be able to protect our trade secrets
or that others will not independently develop substantially equivalent proprietary information and
techniques.
Competition
The development and commercialization of new drugs is highly competitive. Our major competitors are
large pharmaceutical, specialty pharmaceutical and biotechnology companies, in the United States,
Canada and abroad. Many oncology drugs in clinical trials are being developed for the four primary
cancer indications: lung, breast, colorectal, and prostate cancer. Certain of these drugs are, like
OGX-011, OGX-427, and OGX-225, designed to interfere with mechanisms potentially involved with
treatment resistance. If new drugs targeting mechanisms of treatment resistance are approved for
sale for the indications that we are targeting in advance of our product candidates, or even after
their commercialization, it may reduce the markets interest in its product candidates. We are
aware of several other companies developing therapeutics, whether antisense or otherwise, that seek
to promote tumor cell death by inhibiting proteins believed to promote cell survival. Our
competitors may seek to identify gene sequences, protein targets or antisense chemistry different
from ours, and outside the scope of our intellectual property protection, to develop antisense
therapeutics that serve the same function as its product candidates. Our competitors may also seek
to use mechanisms other than antisense to inhibit the proteins that our product candidates are
designed to inhibit the production of.
23
Many of our existing and potential competitors have substantially greater financial resources and
expertise in manufacturing, developing products, conducting clinical trials, obtaining regulatory
approvals, and marketing than us. These entities also compete with us in recruiting and retaining
qualified scientific and management personnel, as well as in acquiring products and technologies
complementary to its programs. Standard treatments vary considerably by cancer indication, and new
drugs may be more effective in treating one cancer indication than another. In addition, it must be
recognized that cancer is a difficult disease to treat and it is likely that no one therapeutic
will replace all other therapies in any particular indication. Therapeutic strategies for treating
cancer are increasingly focused on combining a number of drugs in order to yield the best results.
Since OGX-011 and OGX-427 are intended to be used in multiple cancer indications and target the
tumors adaptive survival mechanisms, these drugs will potentially be synergistic with many new and
currently marketed therapies.
Our ability to compete successfully will depend largely on our ability and, where applicable, the
ability of our collaborators to:
| |
|
|
establish that our product candidates are well tolerated and result in a clinical
benefit when administered to cancer patients; |
| |
|
|
advance the development of our lead programs, including the enrollment of patients for
our clinical trials; |
| |
|
|
gain regulatory approval for our product candidates in their respective first
indications as well as expand into additional indications; |
| |
|
|
commercialize our lead product candidates successfully, including convincing
physicians, insurers and other third-party payors of the advantages of its products, when
and if it has any, over current therapies; |
| |
|
|
obtain intellectual property protection and protect the exclusivity for our product
candidates and products, when and if it has any; and |
| |
|
|
acquire other product candidates to expand our pipeline. |
Trademarks
We own two approved Canadian trademarks: OncoGenex and Bringing Hope to Life. We have registered
corresponding trademark Bringing Hope to Life in the U. S., and applied for OncoGenex in that
jurisdiction. We are aware of a company called Tikvah Therapeutics of Atlanta, Georgia, which has
filed Bringing Hope to Life for different goods and services on an intent-to-use basis. OncoGenex
and Tikvah have agreed not to oppose or prevent the other from establishing their respective marks
for their respective goods.
Registrations and applications relating to the SONUS and TOCOSOL trademarks are no longer being
maintained.
There can be no assurance that the registered or unregistered trademarks or trade names of our
Company will not infringe upon third party rights or will be acceptable to regulatory agencies.
Employees
We have a total of 26 employees; one part-time and 25 full-time. In our Vancouver office, we have
10 full-time employees and one part-time employee, three of whom are engaged in clinical
development and regulatory affairs and seven of whom are engaged in administration, business
development, accounting and finance. In our Bothell office, we have 15 full-time employees, with
all 15 engaged in clinical and regulatory affairs.
24
All of our employees have entered into non-disclosure agreements regarding our intellectual
property, trade secrets and other confidential information. None of our employees are represented
by a labor union or covered by a collective bargaining agreement, nor have we experienced any work
stoppages. We believe that we maintain satisfactory relations with our employees.
From time to time, OncoGenex also uses outside consultants to provide advice on its clinical
development plans, research programs, administration and potential acquisitions of new
technologies.
Company Information
The Company was incorporated in the state of California in October 1991 and subsequently
reorganized as a Delaware corporation in September 2005. Our principal executive offices are
located 1522 217th Place SE, Suite 1090, Bothell, Washington 98021, and its telephone
number is (425) 686-1500.
On August 21, 2008, pursuant to the Arrangement, OncoGenex Technologies Inc. became a wholly-owned
subsidiary of the Company. OncoGenex Technologies was incorporated under the federal laws of Canada
in May 2000. OncoGenex, Inc., the subsidiary of OncoGenex Technologies, was incorporated under the
laws of Washington in August 2005 and was dissolved pursuant to Articles of Dissolution filed on
July 1, 2009.
Available Information
We maintain a website at http://www.oncogenex.com. The information contained on or accessible
through our website is not part of this Annual Report on Form 10-K. Our Annual Report on Form 10-K,
Quarterly Reports on Form 10-Q, Current Reports on Form 8-K and amendments to reports filed or
furnished pursuant to Sections 13(a) and 15(d) of the Securities Exchange Act of 1934, as amended
(the Exchange Act), are available free of charge on our website as soon as reasonably practicable
after we electronically file such reports with, or furnish those reports to, the Securities and
Exchange Commission (the SEC).
25
ITEM 1A. RISK FACTORS
Risks Related to Our Business
Investing in our common shares involves a high degree of risk. You should consider carefully the
risks and
uncertainties described below, together with all of the other information contained in this Form
10-K, before deciding to invest in our common shares. If any of the following risks materialize,
our business, financial condition, results of operation and future prospects will likely be
materially and adversely affected. In that event, the market price of our common shares could
decline and you could lose all or part of your investment.
We have a limited operating history, have incurred losses since inception and anticipate that we
will continue to incur losses for the foreseeable future. We have never had any products available
for commercial sale and we may never achieve or sustain profitability.
We are a clinical-stage biopharmaceutical company with a limited operating history. We are not
profitable and have incurred losses in each year since our inception. We have never had any
products available for commercial sale and we have not generated any revenue from product sales. We
do not anticipate that we will generate revenue from the sale of products in the foreseeable
future. Our only revenue to date has been collaboration revenue under our Collaboration Agreement
with Teva. We have not yet submitted any products for approval by regulatory authorities. We
continue to incur research and development and general and administrative expenses related to our
operations. We expect to continue to incur losses for the foreseeable future, and we expect these
losses to increase as we continue our research activities and conduct development of, and seek
regulatory approvals for, our product candidates, and prepare for and begin to commercialize any
approved products. If our product candidates fail in clinical trials or do not gain regulatory
approval, or if our product candidates do not achieve market acceptance, we may never become
profitable. Even if we achieve profitability in the future, we may not be able to sustain
profitability in subsequent periods.
Because we depend on financing from third parties for our operations, our business may fail if such
financing becomes unavailable or is offered on commercially unreasonable terms.
To date, we have financed our operations primarily through the sale of our equity securities and
from the upfront payment we received pursuant to our Collaboration Agreement with Teva. We believe
that our existing capital resources and interest thereon, including the upfront payment we received
from Teva in December 2009, will be sufficient to meet our current operating requirements into
2012 and expect that both phase 3 prostate cancer trials will be fully accrued by this time. However, if our Collaboration Agreement with Teva were to be terminated or if Teva failed to
fulfill its obligations thereunder, or if we change our development plans, acquire rights to new
product candidates or cannot find third party collaborators for our other product candidates, we
may need additional capital sooner than we expect. Our future capital requirements will depend on
many factors, including without limitation:
| |
|
|
maintaining our partnership with Teva and Tevas ongoing commitment to develop OGX-011
in a timely fashion; |
| |
|
|
the scope and results of our preclinical and clinical trials; |
| |
|
|
whether we experience delays in our preclinical and clinical development programs, or
slower than anticipated product development; |
| |
|
|
whether opportunities to acquire additional product candidates arise and the costs of
acquiring and developing those product candidates; |
| |
|
|
whether we are able to enter into additional third party collaborative partnerships to
develop and/or commercialize any of our other product candidates on terms that are
acceptable to us; |
| |
|
|
the timing and requirements of, and the costs involved in, conducting studies required
to obtain regulatory approvals for our product candidates from the FDA and comparable
foreign regulatory agencies; |
| |
|
|
the availability of third parties to perform the key development tasks for our product
candidates, including conducting preclinical and clinical studies and manufacturing our
product candidates to be tested in those studies and the associated costs of those
services; and |
| |
|
|
the costs involved in preparing, filing, prosecuting, maintaining, defending the
validity of and enforcing, patent claims and other costs related to patent rights and
other intellectual property rights, including litigation costs and the results of such
litigation. |
26
If we are unable to raise funds on acceptable terms when it becomes necessary to do so, we may not
be able to continue developing our product candidates, acquire or develop additional product
candidates or respond to competitive pressures or unanticipated requirements. For these reasons,
any inability to raise additional capital when we require it could have a materially adverse effect
on our business.
We are dependent upon our collaborative relationship with Teva to further develop and commercialize
OGX-011, and if our relationship were not to be successful or were to be terminated, we may not be
able to effectively develop and/or commercialize OGX-011, which would have a material adverse
effect on the Company.
Under our Collaboration Agreement with Teva, we rely heavily on Teva to collaborate with us in
respect of the development and global commercialization of OGX-011. Furthermore, under the
Collaboration Agreement, we and Teva must agree on any changes to the Clinical Development Plan for
OGX-011. As a result of our dependence on our relationship with Teva, the eventual success or
commercial viability of OGX-011 is largely beyond our control. The financial returns to us, if
any, under our Collaboration Agreement with Teva depend in large part on the achievement of
development and commercialization milestones, plus a share of any revenues from sales. Therefore,
our success, and any associated financial returns to us and our investors, will depend in large in
part on the performance of Teva under the agreement.
We are subject to a number of additional specific risks associated with our dependence on our
collaborative relationship with Teva, including:
| |
|
|
adverse decisions by Teva or the Joint Steering Committee regarding the development and
commercialization of OGX-011; |
| |
|
|
possible disagreements as to the timing, nature and extent of our development plans,
including clinical trials or regulatory approval strategy; |
| |
|
|
loss of significant rights if we fail to meet our obligations under the Collaboration
Agreement; |
| |
|
|
our limited control over clinical trials with respect to OGX-011; |
| |
|
|
changes in key management personnel at Teva that are members of the Joint Steering
Committee; and |
| |
|
|
possible disagreements with Teva regarding the Collaboration Agreement or ownership of
proprietary rights. |
If we and Teva are unable to reach an agreement under the Clinical Development Plan, or if either
we or Teva fail to perform our respective obligations or effectively manage our relationship, such
occurrence could cause clinical trial, regulatory approval or development progress to be
significantly delayed or halted, could result in costly or time consuming litigation or arbitration
and could have a material adverse effect on our business.
If Tevas business strategy changes, it may adversely affect the development and commercialization
of OGX-011.
Teva may change its business strategy. Decisions by Teva to either reduce or eliminate its
participation in the oncology field, to emphasize other competitive agents currently in its
portfolio, or to add additional competitive agents to its portfolio, could result in a decision to
terminate the Collaboration Agreement. In the event of termination of the Collaboration Agreement,
among other things, we would have responsibility for paying for any remaining costs of all three
phase 3 clinical trials. Any such termination could adversely affect the timing and extent of development and commercialization activities, which could cause
significant delays and funding shortfalls impacting those activities and seriously harm our
business.
27
We are highly dependent on the success of our lead product candidate, OGX-011, and we cannot give
any assurance that OGX-011 or any of our other product candidates will receive regulatory approval.
OGX-011 has been evaluated in five phase 2 clinical trials, and results for these trials were
previously disclosed. If competitive products developed by third parties show significant benefit
in the cancer indications in which we are developing our product candidates, any planned supportive
or primary registration trials may be delayed, altered or not initiated and OGX-011 may never
receive regulatory approval. In order to market OGX-011, we and Teva must, among other things,
conduct additional clinical trials, including phase 3 or registration clinical trials, to
demonstrate safety and efficacy. We are intending to initiate two registration trials in 2010
involving OGX-011, but we have not initiated any registration clinical trials with any of our
product candidates to date. OGX-427 and SN2310 have been evaluated in humans, though we have very
limited safety data and have not yet established efficacy in humans. We have completed enrollment
in the phase 1 clinical trial of SN2310 and the dose limiting toxicity that defined a maximum
tolerated dose in this heavily pretreated patient population, as expected, was significant
neutropenia. Additional clinical trials will be required with SN2310 to establish the safety and
efficacy of this product candidate. Neither OGX-225 nor CSP-9222 have yet been tested in humans.
Our pre-clinical testing of these product candidates may not be successful and we may be unable to
initiate clinical evaluation of them. Our clinical development programs for our product candidates
may not receive regulatory approval either if such product candidates fail to demonstrate that they
are safe and effective in clinical trials and consequently fail to obtain necessary approvals from
the FDA, or similar non-U.S. regulatory agencies, or if we have inadequate financial or other
resources to advance these product candidates through the clinical trial process. Any failure to
obtain regulatory approval of OGX-011 or our other product candidates would have a material and
adverse impact on our business.
Clinical trials may not demonstrate a clinical benefit of our product candidates.
Positive results from pre-clinical studies and early clinical trials, including those results from
the OGX-011 clinical trials conducted to date, should not be relied upon as evidence that
later-stage or large-scale clinical trials will succeed. We will be required to demonstrate with
substantial evidence through well-controlled clinical trials that our product candidates are safe
and effective for use in a diverse population before we can seek regulatory approvals for their
commercial sale. Success in early clinical trials does not mean that future clinical trials will be
successful because product candidates in later-stage clinical trials may fail to demonstrate
sufficient safety and efficacy to the satisfaction of the FDA and other non-U.S. regulatory
authorities despite having progressed through initial clinical trials.
Even after the completion of our planned phase 3 clinical trials, the FDA or other non-U.S.
regulatory authorities may disagree with our clinical trial design and our interpretation of data,
and may require us to conduct additional clinical trials to demonstrate the efficacy of our product
candidates.
28
Our clinical trials may be suspended or terminated at any time, including by the FDA, other
regulatory authorities, the Institutional Review Board (IRB) overseeing the clinical trial at
issue, any of our clinical trial sites with respect to that site, by Teva in the case of OGX-011,
or by us. Any failure or significant delay in completing clinical trials for our product candidates
could materially harm our financial results and the commercial prospects for our product
candidates.
We do not know whether any of our currently planned future clinical trials for OGX-011 or OGX-427
will proceed or be completed on schedule, or at all, or, with respect our current portfolio of
product candidates, whether we will be able to initiate any future pre-clinical studies or clinical
trials, as applicable, beyond those currently planned. The completion or commencement of future
pre-clinical studies or clinical trials could be substantially delayed or prevented by several
factors, including:
| |
|
|
decrease in Tevas level of focus and efforts to develop OGX-011; |
| |
|
|
delay or failure to obtain sufficient manufacturing supply of OGX-011; |
| |
|
|
delay or failure to obtain acceptance from the FDAs of Avecia as our
manufacturer for OGX-011; |
| |
|
|
limited number of, and competition for, suitable patients with the
particular types of cancer required for enrollment in our clinical
trials; |
| |
|
|
limited number of, and competition for, suitable sites to conduct clinical trials; |
| |
|
|
delay or failure to obtain required future additional funding, when
needed, through private or public offerings of our equity securities,
debt financings, or executing a licensing, partnership or
collaboration agreement with a third party for any of our product
candidates; |
| |
|
|
introduction of new product candidates to the market in therapeutic
areas similar to those which we are developing our product candidates; |
| |
|
|
concurrent evaluation of new investigational product candidates in
therapeutic areas similar to those which we are developing our product
candidates; |
| |
|
|
delay or failure to obtain the FDAs or non-U.S. regulatory agencies
approval or agreement to commence a clinical trial, including our
phase 3 or registration clinical trials under a Special Protocol
Assessment; |
| |
|
|
delay or failure to obtain sufficient supplies of the product candidate for its clinical trials; |
| |
|
|
delay or failure to reach agreement on acceptable clinical trial
agreement terms or clinical trial protocols with prospective sites or
investigators; and |
| |
|
|
delay or failure to obtain the approval of the IRB to conduct a
clinical trial at a prospective site. |
The completion of our clinical trials currently in progress could also be substantially delayed or
prevented by several factors, including:
| |
|
|
slower than expected rates of patient recruitment and enrollment; |
| |
|
|
failure of patients to complete the clinical trial; |
| |
|
|
unforeseen safety issues; |
| |
|
|
lack of efficacy evidenced during clinical trials; |
| |
|
|
termination of our clinical trials by one or more clinical trial sites; |
| |
|
|
inability or unwillingness of patients or medical investigators to
follow its clinical trial protocols; |
| |
|
|
inability to monitor patients adequately during or after treatment; |
| |
| |
|
|
introduction of competitive products that may impede its ability to
retain patients in its clinical trials; and |
| |
|
|
delay or failure to obtain future additional funding through private
or public offerings of our equity securities, debt financings, or
executing a licensing, partnership or collaboration agreement with a
third party for any of our product candidates in the event of material
unforeseen costs relating to our clinical trials currently in
progress. |
29
Our clinical trials may be suspended or terminated at any time by the FDA, other regulatory
authorities, the IRB overseeing the clinical trial at issue, any of our clinical trial sites with
respect to that site, or us. Any failure or significant delay in completing clinical trials for our
product candidates could materially harm our financial results and the commercial prospects for our
product candidates.
Our product candidates may cause undesirable and potentially serious side effects during clinical
trials that could delay or prevent their regulatory approval or commercialization.
OGX-011 has been administered to 294 patients with various types of cancer.
Some of the patients experienced various adverse events, the majority of which are associated with
other treatments in the protocol and the disease. The majority of adverse events were mild and the
most common adverse events associated with OGX-011 consisted of flu-like symptoms. Of the moderate
and severe adverse events associated with OGX-011, neutropenia, vomiting, diarrhea, and difficulty
breathing (also known as dyspnea) were the most common events, occurring in > 2% of patients.
As of February 12, 2010, OGX-427 has been administered to 53 patients with various types of cancer
in a phase 1 clinical trial. Enrollment in the OGX-427 monotherapy aspect of the phase 1 clinical
trial is complete and dose-limiting toxicity was not reached at the highest doses evaluated.
Enrollment in the OGX-427 in combination with docetaxel aspect of the clinical trial is ongoing. Of
the 41 patients presented at ASCO 2009, all patients experienced adverse events, the majority of
which were unrelated to OGX-427. Of the adverse events associated with OGX-427, the majority of
adverse events were mild and the most common adverse events consisted of flu-like symptoms,
infusion-related reactions, pruritus and flushing. Serious adverse events have been reported for
seventeen patients (50%). The serious adverse events were unrelated to OGX-427 administration for
14 patients and associated with OGX-427 administration for 3 patients. The events that were
associated with OGX-427 administration were elevated creatinine in two patients, an indicator of
kidney function, and rigors and chills in one patient.
SN2310 has been administered to 26 patients with various types of cancer in a phase 1 clinical
trial. Enrollment for this clinical trial has been completed. Some of the patients experienced
adverse events, which were considered unrelated to study drug and attributed to underlying disease.
Of the adverse events associated with SN2310, most were mild and the most common events were
nausea, diarrhea, vomiting and fatigue. Mild to moderate reactions (back/chest pain, flushing) have
been observed during infusions. Significant neutropenia has occurred in some patients and was the
dose-limiting toxicity observed, sometimes associated with fever or septicemia.
Since patients in our clinical trials have advanced stages of cancer, we expect that additional
adverse events, including serious adverse events, will occur.
Undesirable side effects caused by any of our product candidates could cause us or regulatory
authorities to interrupt, delay or halt clinical trials and could result in the denial of
regulatory approval by the FDA or non-U.S. regulatory authorities for any or all targeted
indications. This, in turn, could prevent us from commercializing our product candidates and
generating revenues from their sale. In addition, if our product candidates receive marketing
approval and we or others later identify undesirable side effects caused by the product:
| |
|
|
Teva may elect to terminate the ongoing clinical trials and cease development of OGX-011; |
| |
|
|
regulatory authorities may withdraw their approval of the product; |
30
| |
|
|
we may be required to recall the product, change the way the product
is administered, conduct additional clinical trials or change the
labeling of the product; |
| |
|
|
a product may become less competitive and product sales may decrease; or |
| |
|
|
our reputation may suffer. |
Any one or a combination of these events could prevent us from achieving or maintaining market
acceptance of the affected product or could substantially increase the costs and expenses of
commercializing the product, which in turn could delay or prevent us from generating significant
revenues from the sale of the product.
Recent events have raised questions about the safety of marketed drugs and may result in increased
cautiousness by the FDA in reviewing new drugs based on safety, efficacy or other regulatory
considerations and may result in significant delays in obtaining regulatory approvals, additional
clinical trials being required, or more stringent product labeling requirements. Any delay in
obtaining, or inability to obtain, applicable regulatory approvals, would prevent us from
commercializing its product candidates.
Even if we or Teva receive regulatory approval to market our product candidates, the market may not
be receptive to our products.
Even if our product candidates obtain regulatory approval, resulting products may not gain market
acceptance among physicians, patients, health care payors and/or the medical community. We believe
that the degree of market acceptance will depend on a number of factors, including:
| |
|
|
timing of market introduction of competitive products; |
| |
|
|
safety and efficacy of our products; |
| |
|
|
prevalence and severity of any side effects; |
| |
|
|
potential advantages or disadvantages over alternative treatments; |
| |
|
|
strength of marketing and distribution support; |
| |
|
|
price of our products, both in absolute terms and relative to alternative treatments;
and |
| |
|
|
availability of coverage and reimbursement from government and other third-party
payors. |
If our future product candidates fail to achieve market acceptance, we may not be able to generate
significant revenue or achieve or sustain profitability.
We may not be able to negotiate the exit or sublease of excess office and laboratory space
currently leased in Bothell, Washington, on terms acceptable to us or at all.
Prior to the Arrangement, Sonus entered into a non-cancellable lease arrangement for office and
laboratory space located in Bothell, Washington, which is considered to be in excess of our current
requirements. We have been seeking to exit or sublease this excess space. To date, we have not
entered into any agreement for the exit or sublease of this space, or identified which transactions
or transaction structures would most benefit shareholders. The goal of minimizing future lease
expenditures will impact any decisions we make regarding specific deal structures or transactions
into which we may enter. We can provide no assurances
that we will be able to negotiate the exit or sublease of this space, on terms acceptable to us or
at all or on terms which meet our or our shareholders expectations.
31
Although we have entered into a Collaboration Agreement with Teva for OGX-011, we have not yet
partnered with third party collaborators with respect to any of our other product candidates, and
we cannot control whether we will be able to do so on favorable terms or at all.
Our business strategy relies in part on potentially partnering successful product candidates with
larger companies to complement our internal development and commercialization efforts. While we
have successfully entered into a Collaboration Agreement with Teva with respect to OGX-011, it may
be difficult for us to find third parties that are willing to enter into a collaboration on
acceptable economic terms or at all with respect to our other product candidates. We also will be
competing with many other companies as we seek partners for our other product candidates and we may
not be able to compete successfully against those other companies. If we are not able to enter
into collaboration arrangements for our other product candidates, we would be required to undertake
and fund further development, clinical trials, manufacturing and commercialization activities
solely at our own expense and risk. If we are unable to finance and/or successfully execute those
expensive activities, our business could be materially and adversely effected.
If our competitors develop and market products that are more effective, safer or less expensive
than our future product candidates, our clinical trials and commercial opportunities will be
negatively impacted.
The life sciences industry is highly competitive, and we face significant competition from many
pharmaceutical, biopharmaceutical and biotechnology companies that are researching and marketing
products designed to address cancer indications for which we are currently developing products or
for which we may develop products in the future. We are aware of several other companies which are
developing therapeutics that seek to promote tumor cell death by inhibiting proteins believed to
promote cell survival. Any products we may develop in the future are also likely to face
competition from other drugs and therapies. Many of our competitors have significantly greater
financial, manufacturing, marketing and drug development resources than we do. Large pharmaceutical
companies, in particular, have extensive experience in clinical testing and in obtaining regulatory
approvals for drugs. These companies also have significantly greater research and marketing
capabilities than we do. In addition, many universities and private and public research institutes
are, or may become, active in cancer research, the products of which may be in direct competition
with ours. If our competitors market products that are more effective, safer or less expensive than
our future product candidates, if any, or that reach the market sooner than our future product
candidates, if any, we may not achieve commercial success.
If new therapies become broadly used, we may need to conduct clinical trials of our product
candidates in combination with these new therapies to demonstrate safety and efficacy of the
combination. Additional trials will delay the development of our product candidates and increase
our costs. The failure of certain of our product candidates to work in combination with these new
therapies would have an adverse effect on our business.
Our intention is to combine certain of our product candidates with therapies that are broadly used
by clinicians and considered highly effective. As new therapies are developed, we will need to
assess these therapies to determine whether to conduct clinical trials of our product candidates in
combination with them to demonstrate safety and efficacy of the combination. If we determine that
it is appropriate to conduct additional clinical trials of our product candidates in combination
with these new therapies, the development of our product candidates will be delayed and our costs
will be increased. If these clinical trials generate safety concerns or lack of efficacy, our
business would be adversely affected.
If our product candidates become approved in combination with a specific therapy that is broadly
used and that therapy becomes displaced by another product, the market for our product candidate
may decrease.
32
If we were to be successfully sued related to our products or operations, we could face substantial
liabilities that may exceed our resources.
We may be held liable if any of our products or operations cause injury or death or are found
otherwise unsuitable during product testing, manufacturing, marketing or sale. These risks are
inherent in the development of pharmaceutical products. We currently maintain Commercial General
and Umbrella Liability policies with combined limits of $10 million per occurrence and in the
aggregate and in addition, a $10 million per claim and annual aggregate product liability insurance
policy related to our clinical trials consistent with industry standards. When necessary for our
products, we intend to obtain additional product liability insurance. Insurance coverage may be
prohibitively expensive, may not fully cover potential liabilities or may not be available in the
future. Inability to obtain sufficient insurance coverage at an acceptable cost or otherwise to
protect against potential product liability claims could prevent or inhibit the commercialization
of our products. If we were to be sued for any injury caused by or associated with our products or
operations, the litigation could consume substantial time and attention of our management, and the
resulting liability could exceed our total assets.
If we fail to acquire and develop products or product candidates at all or on commercially
reasonable terms, we may be unable to grow our business.
We currently do not have internal discovery capabilities and are dependent upon pharmaceutical and
biotechnology companies and other researchers to sell or license products or product candidates to
us. To date, three of our product candidates have been derived from
technologies discovered by the Vancouver Prostate Centre and licensed to us by UBC and one candidate has been in-licensed from Bayer. We
intend to continue to rely on the Vancouver Prostate Centre, UBC and other research institutions and other
biotechnology or pharmaceutical companies as sources of product candidates. We cannot guarantee
that the Vancouver Prostate Centre or UBC will continue to develop new product candidate opportunities, that
we will continue to have access to such opportunities or that we will be able to purchase or
license these product candidates on commercially reasonable terms, or at all. If we are unable to
purchase or license new product candidates from the Vancouver Prostate Centre or UBC, we will be required to
identify alternative sources of product candidates.
The success of our product pipeline strategy depends upon our ability to identify, select and
acquire pharmaceutical product candidates. Proposing, negotiating and implementing an economically
viable product acquisition or license is a lengthy and complex process. We compete for partnering
arrangements and license agreements with pharmaceutical and biotechnology companies and academic
research institutions. Our competitors may have stronger relationships with third parties with whom
we are interested in collaborating and/or may have more established histories of developing and
commercializing products. As a result, our competitors may have a competitive advantage in entering
into partnering arrangements with such third parties. In addition, even if we find promising
product candidates, and generate interest in a partnering or strategic arrangement to acquire such
product candidates, we may not be able to acquire rights to additional product candidates or
approved products on terms that we find acceptable, or at all. If we fail to acquire and develop
product candidates from others, we may be unable to grow our business.
We expect that any product candidate that we acquire rights to will require additional development
efforts prior to commercial sale, including extensive clinical evaluation and approval by the FDA
and non-U.S. regulatory authorities. All product candidates are subject to the risks of failure
inherent in pharmaceutical product development, including the possibility that the product
candidate will not be shown to be sufficiently safe and effective for approval by regulatory
authorities. Even if the product candidates are approved, we cannot be sure that we would be
capable of economically feasible production or commercial success.
33
We will need to retain additional personnel and expand our other resources in order to complete
development of OGX-011, promote OGX-011 in the event we exercise our co-promotion option and
develop our other product candidates. If we fail to effectively expand our operations, including
attracting and retaining key management and scientific personnel, we may be unable to successfully
develop or commercialize our product candidates and our business may be materially adversely
affected.
We will need to expand and effectively manage our managerial, operational, financial, development
and other resources in order to successfully pursue our research, development and commercialization
efforts for our existing and future product candidates. Our success depends on our continued
ability to attract, retain and motivate highly qualified personnel, including management,
pre-clinical and clinical personnel, including our executive officers Scott Cormack and Cindy
Jacobs, and to recruit and retain a new Chief Financial officer. Currently, Cameron Lawrence, the
Director of Financial Reporting, is our interim principal financial officer. We are in the process
of searching for executive talent to fill the Chief Financial Officer
position on a permanent basis.
In addition, although we have entered into employment agreements with each of Mr. Cormack and
Dr. Jacobs, such agreements permit the executive to terminate his or her employment with us at any
time, subject to providing us with advance written notice.
With respect to OGX-011 specifically, we will need to hire additional clinical development
personnel in order to perform our responsibilities under the Clinical Development Plan with Teva.
In addition, should OGX-011 receive marketing approval in the United States and Canada, or should
we exercise our co-promotion option, which we do not anticipate having the funds to do, we would
need to hire a substantial number of specialized personnel, including field based medical affairs
representatives. In turn, we would need to increase our administrative headcount to support such
expanded development and commercialization operations with respect to our product candidates. Our
ability to attract and retain qualified personnel in the future is subject to intense competition
for qualified personnel among biotechnology, pharmaceutical and other businesses and our current
financial position. The loss of the services of any of our senior management, or our inability
to recruit a new Chief Financial Officer, could delay or prevent the development and
commercialization of our product candidates, or have other adverse effects on our business for an
indefinite term. In particular, if we lose any members of our current senior management team, we
may not be able to find suitable replacements in a timely fashion or at all and our business may be
harmed as a result. Among other things, if any of such events were to occur, we may not be able to
comply with our contractual obligations to Teva under our Collaboration Agreement or advance our
product candidates, which could have a material adverse effect on our business.
We have scientific and clinical advisors who assist us in formulating our development and clinical
strategies. These advisors are not employees of the Company and may have commitments to, or
consulting or advisory contracts with, other entities that may limit their availability to us. In
addition, our advisors may have arrangements with other companies to assist those companies in
developing products or technologies that may compete with ours.
We may encounter difficulties in managing our expected growth and in expanding our operations
successfully.
As we advance our product candidates OGX-427, OGX-225, and CSP-9222 through development and
clinical trials, we will need to develop or expand our development, regulatory, manufacturing,
marketing and sales capabilities or contract with third parties to provide these capabilities for
us. Maintaining additional relationships and managing our future growth will impose significant
added responsibilities on members of our management. We must be able to: manage our development
efforts effectively; manage our clinical trials effectively; hire, train and integrate additional
management, development, administrative and sales and marketing personnel; improve our managerial,
development, operational and finance systems; and expand our facilities, all of which may impose a
strain on our administrative and operational infrastructure.
Under the Collaboration Agreement, Teva is responsible for the commercialization costs associated
with OGX-011; however, if we were to exercise our co-promotion option, which we do not anticipate
having sufficient funds to do, we would need to expand our marketing and sales capabilities. In
addition, as we have primary responsibility for the oversight of the second-line trial in CRPC, we
must be able to manage our development responsibilities effectively, which may impose a strain on
our administrative and operational infrastructure.
34
Furthermore, we may acquire additional businesses, products or product candidates that complement
or augment our existing business. Integrating any newly acquired business, product or product
candidate could be expensive and time-consuming. We may not be able to integrate any acquired
business, product or product candidate successfully or operate any acquired business profitably.
Our future financial performance will depend, in part, on our ability to manage any future growth
effectively and our ability to integrate any acquired businesses. We may not be able to accomplish
these tasks, and our failure to accomplish any of them could prevent us from successfully growing
the Company.
We may need to further develop our financial and reporting processes, procedures and controls to
support our anticipated growth.
To manage the anticipated growth of our operations and personnel, we may be required to improve
existing, or implement new, operational and financial systems, processes and procedures, and to
expand, train and manage our employee base. Our current and planned systems, procedures and
controls may not be adequate to support our future operations.
We may be adversely impacted if our controls over external financial reporting fail or are
circumvented.
We regularly review and update our internal controls, disclosure controls and procedures, and
corporate governance policies. In addition, we are required under the Sarbanes Oxley Act of 2002 to
report annually on our internal control over financial reporting. If it were to be determined that
our internal control over financial reporting is not effective, such shortcoming could have an
adverse effect on our business and financial results and the price of our common stock could be
negatively affected. This reporting requirement could also make it more difficult or more costly
for us to obtain certain types of insurance, including director and officer liability insurance,
and we may be forced to accept reduced policy limits and coverage or incur substantially higher
costs to obtain the same or similar coverage. Any system of internal controls, however well
designed and operated, is based in part on certain assumptions and can provide only reasonable, not
absolute, assurances that the objectives of the system are met. Any failure or circumvention of the
controls and procedures or failure to comply with regulation concerning control and procedures
could have a material effect on our business, results of operation and financial condition. Any of
these events could result in an adverse reaction in the financial marketplace due to a loss of
investor confidence in the reliability of our financial statements, which ultimately could
negatively impact the market price of our shares, increase the volatility of our stock price and
adversely affect our ability to raise additional funding. The impact of these events could also
make it more difficult for us to attract and retain qualified persons to serve on our board of
directors, our board committees and as executive officers.
We rely, in part, on third parties to conduct clinical trials for our product candidates and plan
to rely on third parties to conduct future clinical trials. If these third parties do not
successfully carry out their contractual duties or meet expected deadlines, we may be unable to
obtain regulatory approval for or commercialize our current and future product candidates.
To implement our product development strategies, we rely on third parties, such as collaborators,
contract research organizations, medical institutions, clinical investigators and contract
laboratories, to conduct our clinical trials of our product candidates. In particular, we will
have limited control over the two OGX-O11 phase 3 trials over which Teva will have primary
oversight. Although we rely on third parties to conduct our clinical trials, we are responsible
for ensuring that each of our clinical trials is conducted in accordance with our investigational
plan and protocol. Moreover, the FDA and non-U.S. regulatory authorities require us to comply with
regulations and standards, commonly referred to as Good Clinical Practices (GCPs) for conducting,
monitoring, recording and reporting the results of clinical trials to ensure that the data and
results are scientifically credible and accurate and that the clinical trial subjects are
adequately informed of the potential risks of participating in clinical trials. Our reliance on
third parties does not relieve us of these responsibilities and requirements. If the third parties
conducting our clinical trials do not perform their contractual duties or obligations, do not meet
expected deadlines or need to be replaced, or if the quality or accuracy of the clinical data they
obtain is compromised due to the failure to adhere to GCPs or for any
other reason, we may need to enter into new arrangements with alternative third parties and our
clinical trials may be extended, delayed or terminated. In addition, a failure by such third
parties to perform their obligations in compliance with GCPs may cause our clinical trials to fail
to meet regulatory requirements, which may require us to repeat our clinical trials.
35
We rely on third parties to manufacture and supply our product candidates.
We do not own or operate manufacturing facilities, and we depend on third-party contract
manufacturers for production of our product candidates. We lack the resources and the capability to
manufacture any of our product candidates ourselves. To date, our product candidates have been
manufactured in limited quantities for pre-clinical studies and clinical trials. All active
pharmaceutical ingredient for OGX-011 has been manufactured for us by Isis or Avecia and all drug
product has been manufactured for us by Formatech, Inc. and Pyramid Laboratories, Inc., in each
case pursuant to a purchase order or short-term contract that has been fulfilled. We will need to
obtain additional quantities of OGX-011 to complete our first phase 3 clinical trial.
All active pharmaceutical ingredient for OGX-427 for IND-enabling toxicology studies and initial
clinical trials has been manufactured for us by Avecia and all drug product has been manufactured
for us by Laureate Pharma, Inc., in each case pursuant to a purchase order or short-term contract
that has been fulfilled.
If, in the future, one of our product candidates is approved for commercial sale, we or a
pharmaceutical partner that has licensed such product candidates, will need to manufacture that
product candidate in commercial quantities. We cannot assure you that the third-party manufacturers
with which we have contracted in the past will have sufficient capacity to satisfy our future
manufacturing needs, that we will be able to negotiate additional purchases of active
pharmaceutical ingredient or drug product from these or alternative manufacturers on terms
favorable to us, or at all, or that a pharmaceutical partner that has licensed will have sufficient
capacity or expertise to satisfy future needs.
Third party manufacturers may fail to perform under their contractual obligations, or may fail to
deliver the required commercial quantities of bulk drug substance or finished product on a timely
basis and at commercially reasonable prices. Any performance failure on the part of our contract
manufacturers could delay clinical development or regulatory approval of our product candidates or
commercialization of our future product candidates, depriving us of potential product revenue and
resulting in additional losses. If we are required to identify and qualify an alternate
manufacturer, we may be forced to delay or suspend our clinical trials, regulatory submissions,
required approvals or commercialization of our product candidates, which may cause us to incur
higher costs and could prevent us from commercializing our product candidates successfully. If we
are unable to find one or more replacement manufacturers capable of production at a reasonably
favorable cost, in adequate volumes, of adequate quality, and on a timely basis, we would likely be
unable to meet demand for our product candidates and our clinical trials could be delayed or we
could lose potential revenue. Our ability to replace an existing active pharmaceutical ingredient
manufacturer may be difficult because the number of potential manufacturers is limited to
approximately four manufacturers, and the FDA must inspect any replacement manufacturer and review
information related to product produced at the manufacturer before they can begin manufacturing our
product candidates. It may be difficult or impossible for us to identify and engage a replacement
manufacturer on acceptable terms in a timely manner, or at all. We expect to continue to depend on
third-party contract manufacturers for the foreseeable future.
Our product candidates require precise, high quality manufacturing. Any of our contract
manufacturers will be subject to ongoing periodic unannounced inspection by the FDA and non-U.S.
regulatory authorities to ensure strict compliance with cGMP, and other applicable government
regulations and corresponding standards. If our contract manufacturers fail to achieve and maintain
high manufacturing standards in compliance with cGMP regulations, we may experience manufacturing
errors resulting in patient injury or death, product recalls or withdrawals, delays or
interruptions of production or failures in product testing or delivery, delay or prevention of
filing or approval of marketing applications for our product candidates, cost overruns or other
problems that could seriously harm our business.
Significant scale-up of manufacturing may require additional validation studies, which the FDA must
review and approve. Additionally, any third party manufacturers we retain to manufacture our
product candidates on a commercial scale must pass an FDA pre-approval inspection for conformance
to the cGMPs before we can obtain approval of our product candidates. If we are unable to
successfully increase the manufacturing capacity for a product candidate in conformance with cGMPs,
the regulatory approval or commercial launch of any related products may be delayed or there may be
a shortage in supply.
36
Global credit and financial market conditions could negatively impact the value of our current
portfolio of cash equivalents and investment securities
Our cash and cash equivalents are maintained in highly liquid investments with maturities of 90
days or less at the time of purchase. As of the date of this filing, we are not aware of any
material losses or other significant deterioration in the fair value of our cash equivalents or
investment securities since December 31, 2009. However, no assurance can be given that
deterioration in conditions of the global credit and financial markets would not negatively impact
our current portfolio of cash equivalents and investment securities and, as result, our financial
condition.
Risks Related to Our Intellectual Property
Our proprietary rights may not adequately protect our technologies and product candidates.
Our commercial success will depend on our ability to obtain patents and/or regulatory exclusivity
and maintain adequate protection for our technologies and product candidates in the United States
and other countries. We will be able to protect our proprietary rights from unauthorized use by
third parties only to the extent that our proprietary technologies and future product candidates
are covered by valid and enforceable patents or are effectively maintained as trade secrets.
We and our collaborators, including Teva, intend to apply for additional patents covering both our
technologies and product candidates, as we deem appropriate. However, we or our collaborators may
fail to apply for patents on important technologies or product candidates in a timely fashion, or
at all. Our existing patents and any future patents we or our collaborators obtain may not be
sufficiently broad to prevent others from practicing our technologies or from developing competing
products and technologies. In addition, we do not always control the patent prosecution of subject
matter that we license from others. Accordingly, we are sometimes unable to exercise a significant
degree of control over such intellectual property as we would over our own. Moreover, the patent
positions of biopharmaceutical companies are highly uncertain and involve complex legal and factual
questions for which important legal principles remain unresolved. As a result, the validity and
enforceability of our patents cannot be predicted with certainty. In addition, we cannot guarantee
that:
| |
|
|
we or our licensors were the first to make the inventions covered by
each of our issued patents and pending patent applications; |
| |
|
|
we or our licensors were the first to file patent applications for these inventions; |
| |
|
|
others will not independently develop similar or alternative
technologies or duplicate any of our technologies; |
| |
|
|
any of our or our licensors pending patent applications will result in issued patents; |
| |
|
|
any of our or our licensors patents will be valid or enforceable; |
37
| |
|
|
any patents issued to us or our licensors and collaboration partners
will provide us with any competitive advantages, or will not be
challenged by third parties; |
| |
|
|
we will develop additional proprietary technologies that are patentable; or |
| |
|
|
the patents of others will not have an adverse effect on our business. |
The actual protection afforded by a patent varies on a product-by-product basis, from country to
country and depends upon many factors, including the type of patent, the scope of its coverage, the
availability of regulatory related extensions, the availability of legal remedies in a particular
country and the validity and enforceability of the patents. The ability of us or our collaborators
to maintain and solidify our proprietary position for our product candidates will depend on our
success in obtaining effective claims and enforcing those claims once granted. Our issued patents
and those that may issue in the future, or those licensed to us or our collaborators, may be
challenged, invalidated, unenforceable or circumvented, and the rights granted under any issued
patents may not provide us with proprietary protection or competitive advantages against
competitors with similar products. Due to the extensive amount of time required for the
development, testing and regulatory review of a potential product, it is possible that, before any
of our product candidates can be commercialized, any related patent may expire or remain in force
for only a short period following commercialization, thereby reducing any advantage of the patent.
We and our collaborators, including Teva, also rely on trade secrets to protect some of our
technology, especially where it is believed that patent protection is appropriate or obtainable.
However, trade secrets are difficult to maintain. While we use reasonable efforts to protect our
trade secrets, our or our collaboration partners employees, consultants, contractors or scientific
and other advisors may unintentionally or willfully disclose our proprietary information to
competitors. Enforcement of claims that a third party has illegally obtained and is using trade
secrets is expensive, time consuming and uncertain. In addition, non-U.S. courts are sometimes less
willing than U.S. courts to protect trade secrets. If our competitors independently develop
equivalent knowledge, methods and know-how, we would not be able to assert our trade secrets
against them and our business could be harmed.
We and our collaborators, including Teva, may not be able to protect our intellectual property
rights throughout the world.
Filing, prosecuting and defending patents on all of our product candidates and products, when and
if we have any, in every jurisdiction would be prohibitively expensive. Competitors may use our
technologies in jurisdictions where we or our licensors have not obtained patent protection to
develop their own products. These products may compete with our products, when and if we have any,
and may not be covered by any of our or our licensors patent claims or other intellectual property
rights.
The laws of some non-U.S. countries do not protect intellectual property rights to the same extent
as the laws of the United States, and many companies have encountered significant problems in
protecting and defending such rights in foreign jurisdictions. The legal systems of certain
countries, particularly certain developing countries, do not favor the enforcement of patents and
other intellectual property protection, particularly those relating to biotechnology and/or
pharmaceuticals, which could make it difficult for us to stop the infringement of our patents.
Proceedings to enforce our patent rights in foreign jurisdictions could result in substantial cost
and divert our efforts and attention from other aspects of our business.
The intellectual property protection for our product candidates is dependent on third parties.
With respect to OGX-011, OGX-427 and OGX-225, we have exclusively licensed from UBC certain issued
patents and pending patent applications covering the respective antisense sequences underlying
these product candidates and their commercialization and use and we have licensed from Isis certain
issued patents and pending patent applications directed to product compositions and chemical
modifications used in product candidates for commercialization, use and the manufacturing thereof,
as well as some alternative antisense sequences. We have also received a sublicense from Isis under
certain third party patent portfolios directed to such modifications. We have entered into an
exclusive in-licensing agreement with Bayer for development of caspase activators that are
presently being evaluated in preclinical studies.
38
The patents and pending patent applications underlying our licenses do not cover all potential
product candidates, modifications and uses. In the case of patents and patent applications licensed
from Isis and Bayer, we do not have and have not had any control over the filing, prosecution or
enforcement of these patents or patent applications. We cannot be certain that such prosecution
efforts have been or will be conducted in compliance with applicable laws and regulations or will
result in valid and enforceable patents. We also cannot be assured that our licensors or their
respective licensors will agree to enforce any such patent rights at our request or devote
sufficient efforts to attain a desirable result. Any failure by our licensors or any of their
respective licensing partners to properly protect the intellectual property rights relating to our
product candidates could have a material adverse effect on our financial condition and results of
operation.
We may become involved in disputes with Teva or potential future collaborators over intellectual
property ownership, and publications by our research collaborators and scientific advisors could
impair our ability to obtain patent protection or protect our proprietary information, which, in
either case, could have a significant impact on our business.
Inventions discovered under research, material transfer or other such collaborative agreements,
including our Collaboration Agreement with Teva, may become jointly owned by us and the other party
to such agreements in some cases and the exclusive property of either party in other cases. Under
some circumstances, it may be difficult to determine who owns a particular invention, or whether it
is jointly owned, and disputes could arise regarding ownership of those inventions. These disputes
could be costly and time consuming and an unfavorable outcome could have a significant adverse
effect on our business if we were not able to protect or license rights to these inventions. In
addition, our research collaborators and scientific advisors generally have contractual rights to
publish our data and other proprietary information, subject to our prior review. Publications by
our research collaborators and scientific advisors containing such information, either with our
permission or in contravention of the terms of their agreements with us, may impair our ability to
obtain patent protection or protect our proprietary information, which could significantly harm our
business.
The patent protection for our product candidates or products may expire before we are able to
maximize their commercial value which may subject us to increased competition and reduce or
eliminate our opportunity to generate product revenue.
The patents for our product candidates have varying expiration dates and, when these patents
expire, we may be subject to increased competition and we may not be able to recover our
development costs. For example, the first granted U.S. patent directed to OGX-011 and licensed from
UBC is due to expire in 2021. In some of the larger economic territories, such as the United States
and Europe, patent term extension/restoration may be available to compensate for time taken during
aspects of the product candidates regulatory review. However, we cannot be certain that an
extension will be granted, or if granted, what the applicable time period or the scope of patent
protection afforded during any extended period will be. In addition, even though some regulatory
agencies may provide some other exclusivity for a product candidate under its own laws and
regulations, we may not be able to qualify the product candidate or obtain the exclusive time
period.
If we are unable to obtain patent term extension/restoration or some other exclusivity, we could be
subject to increased competition and our opportunity to establish or maintain product revenue could
be substantially reduced or eliminated. Furthermore, we may not have sufficient time to recover our
development costs prior to the expiration of our U.S. and non-U.S. patents.
39
We may incur substantial costs as a result of litigation or other proceedings relating to patent
and other intellectual property rights and we may be unable to protect our rights to, or use of,
our technology.
If we choose to go to court to stop someone else from using the inventions claimed in our patents
or our licensed patents, that individual or company has the right to ask the court to rule that
these patents are invalid and/or should not be enforced against that third party. These lawsuits are expensive and
would consume time and other resources even if we were successful in stopping the infringement of
these patents. In addition, there is a risk that the court will decide that these patents are
invalid or unenforceable and that we do not have the right to stop the other party from using the
inventions. There is also the risk that, even if the validity or unenforceability of these patents
is upheld, the court will refuse to stop the other party on the grounds that such other partys
activities do not infringe our rights.
If we wish to use the technology or compound claimed in issued and unexpired patents owned by
others, we will need to obtain a license from the owner, enter into litigation to challenge the
validity or enforceability of the patents or incur the risk of litigation in the event that the
owner asserts that we infringed its patents. The failure to obtain a license to technology or the
failure to challenge an issued patent that we may require to discover, develop or commercialize our
product candidates may have a material adverse impact on us.
If a third party asserts that we infringed its patents or other proprietary rights, we could face a
number of risks that could seriously harm our results of operations, financial condition and
competitive position, including:
| |
|
|
patent infringement and other intellectual property claims, which
would be costly and time consuming to defend, whether or not the
claims have merit, and which could delay the regulatory approval
process and divert managements attention from our business; |
| |
| |
|
|
substantial damages for past infringement, which we may have to pay if
a court determines that our product candidates or technologies
infringe a competitors patent or other proprietary rights; |
| |
| |
|
|
a court prohibiting us from selling or licensing our technologies or
future drugs unless the third party licenses its patents or other
proprietary rights to us on commercially reasonable terms, which it is
not required to do; and |
| |
| |
|
|
if a license is available from a third party, we may have to pay
substantial royalties or lump sum payments or grant cross licenses to
our patents or other proprietary rights to obtain that license. |
The biotechnology industry has produced a proliferation of patents, and it is not always clear to
industry participants, including us, which patents cover various types of products or methods of
use. The coverage of patents is subject to interpretation by the courts, and the interpretation is
not always uniform. If we are sued for patent infringement, we would need to demonstrate that our
product candidates or methods of use either do not infringe the patent claims of the relevant
patent, and/or that the patent claims are invalid, and/or the patent is unenforceable and we may
not be able to do this. Proving invalidity, in particular, is difficult since it requires a showing
of clear and convincing evidence to overcome the presumption of validity enjoyed by issued patents.
U.S. patent laws as well as the laws of some foreign jurisdictions provide for provisional rights
in published patent applications beginning on the date of publication, including the right to
obtain reasonable royalties, if a patent subsequently issues and certain other conditions are met.
Because some patent applications in the United States may be maintained in secrecy until the
patents are issued, because patent applications in the United States and many foreign jurisdictions
are typically not published until 18 months after filing, and because publications in the
scientific literature often lag behind actual discoveries, we cannot be certain that others have
not filed patent applications for technology covered by our licensors issued patents or our
pending applications or our licensors pending applications, or that we or our licensors were the
first to invent the technology.
40
Patent applications filed by third parties that cover technology similar to ours may have priority
over our or our licensors patent applications and could further require us to obtain rights to
issued patents covering such technologies. If another party files a United States patent application on an invention
similar to ours, we may elect to participate in or be drawn into an interference proceeding
declared by the U.S. PTO to determine priority of invention in the United States. The costs of these
proceedings could be substantial, and it is possible that such efforts would be unsuccessful,
resulting in a loss of our United States patent position with respect to such inventions.
Some of our competitors may be able to sustain the costs of complex patent litigation more
effectively than we can because they have substantially greater resources. In addition, any
uncertainties resulting from the initiation and continuation of any litigation could have a
material adverse effect on our ability to raise the funds necessary to continue our operations. We
cannot predict whether third parties will assert these claims against us or against the licensors
of technology licensed to us, or whether those claims will harm our business. If we are forced to
defend against these claims, whether they are with or without any merit, whether they are resolved
in favor of or against us or our licensors, we may face costly litigation and diversion of
managements attention and resources. As a result of these disputes, we may have to develop costly
non-infringing technology, or enter into licensing agreements. These agreements, if necessary, may
be unavailable on terms acceptable to us, if at all, which could seriously harm our business or
financial condition.
If we breach any of the agreements under which we license rights to our product candidates or
technology from third parties, we could lose license rights that are important to our business.
Certain of our license agreements may not provide an adequate remedy for their breach by the
licensor.
We license the development and commercialization rights for most of our product candidates,
including OGX-011, OGX-427, OGX-225 and CSP-9222, and we expect to enter into similar licenses in
the future. Under such licenses, we are subject to various obligations such as sublicensing,
royalty and milestone payments, annual maintenance fees, limits on sublicensing, insurance
obligations and the obligation to use commercially reasonable best efforts to develop and exploit
the licensed technology. If we fail to comply with any of these obligations or otherwise breach
these agreements, our licensors may have the right to terminate the license in whole or in part or
to terminate the exclusive nature of the license. Loss of any of these licenses or the exclusivity
rights provided therein could harm our financial condition and operating results. In addition,
certain of our license agreements with UBC eliminate our ability to obtain money damages in respect
of certain claims against UBC.
Under the terms of our Collaboration Agreement with Teva, we are required to use commercially
reasonable efforts to maintain and not to breach in any material manner certain of our third party
license agreements relating to OGX-011. If we breach any of these agreements in a material manner,
we would be in breach of the Collaboration Agreement with Teva, which would allow them to terminate
the Collaboration Agreement.
We may be subject to damages resulting from claims that we, or our employees or consultants, have
wrongfully used or disclosed alleged trade secrets of third parties.
Many of our employees were previously employed, and certain of our consultants are currently
employed, at universities or biotechnology or pharmaceutical companies, including our competitors
or potential competitors. Although we have not received any claim to date, we may be subject to
claims that these employees or consultants or we have inadvertently or otherwise used or disclosed
trade secrets or other proprietary information of these current or former employers. Litigation may
be necessary to defend against these claims. If we fail in defending such claims, in addition to
paying monetary damages, we may lose valuable intellectual property rights or personnel. We may be
subject to claims that employees of our partners or licensors of technology licensed by us have
inadvertently or otherwise used or disclosed trade secrets or other proprietary information of
their former employers. We may become involved in litigation to defend against these claims. If we
fail in defending such claims, in addition to paying monetary damages, we may lose valuable
intellectual property rights or personnel.
41
Risks Related to our Common Stock and Other Securities
If we raise additional financing, the terms of such transactions may cause dilution to existing
shareholders or contain terms that are not favorable to us.
To date, our sources of cash have been limited primarily to proceeds from the private or public
placement of our securities and proceeds from the Collaboration Agreement with Teva. In the future,
we may seek to raise additional financing through private placements or public offerings of our
equity or debt securities. We cannot be certain that additional funding will be available on
acceptable terms, or at all. To the extent that we raise additional funds by issuing equity
securities, our shareholders may experience significant dilution. Any debt financing, if available,
may involve restrictive covenants, such as limitations on our ability to incur additional
indebtedness, limitations on our ability to acquire or license intellectual property rights and
other operating restrictions that could adversely impact our ability to conduct our business.
The price for our common stock is volatile.
The market prices for our common stock and that of emerging growth companies generally have
historically been highly volatile. Future announcements concerning us or our competitors may have a
significant impact on the market price of our common stock.
The stock markets also experience significant price and volume fluctuation unrelated to the
operating performance of particular companies. These market fluctuations may also adversely affect
the market price of our common stock.
An increase in the market price of our common shares, which is uncertain and unpredictable, may be
your sole source of gain from an investment in our common shares. An investment in our common
shares may not be appropriate for investors who require dividend income.
We have never declared or paid cash dividends on our capital stock. We do not anticipate paying any
cash dividends on our capital stock in the foreseeable future. We currently intend to retain all
available funds and any future earnings to fund the development and growth of our business. As a
result, capital appreciation, if any, of our common stock will be your sole source of gain for the
foreseeable future. Accordingly, an investment in our common shares may not be appropriate for
investors who require dividend income.
Anti-takeover provisions in our shareholder rights plan, our constating documents and under
Delaware law could make a third party acquisition of the Company difficult.
We have a shareholder rights plan that may have the effect of discouraging unsolicited takeover
proposals. Specifically, the rights issued under the shareholder rights plan could cause
significant dilution to a person or group that attempts to acquire us on terms not approved in
advance by our board of directors. In addition, our certificate of incorporation and bylaws contain
provisions that may discourage unsolicited takeover proposals that shareholders may consider to be
in their best interests. These provisions include the ability of our board of directors to
designate the terms of and issue new series of preferred stock and the ability of our board of
directors to amend the bylaws without stockholder approval. In addition, as a Delaware corporation,
we are subject to Section 203 of the Delaware General Corporation Law, which generally prohibits a
Delaware corporation from engaging in any business combination with any interested stockholder for
a period of three years following the date that the stockholder became an interested stockholder,
unless certain specific requirements are met as set forth in Section 203. Collectively, these
provisions could make a third party acquisition of the Company difficult or could discourage
transactions that otherwise could involve payment of a premium over prevailing market prices for
our common stock.
42
Risks Related to Our Industry
The regulatory approval process is expensive, time consuming and uncertain and may prevent us from
obtaining approvals for the commercialization of some or all of our product candidates.
The research, testing, manufacturing, labeling, approval, selling, marketing and distribution of
drug products are subject to extensive regulation by the FDA and non-U.S. regulatory authorities,
which regulations differ from country to country. We are not permitted to market our product
candidates in the United States until we receive approval of a NDA, from the FDA. We have not submitted an
application for or received marketing approval for any of our product candidates. Obtaining
approval of an NDA can be a lengthy, expensive and uncertain process. In addition, failure to
comply with FDA, non-U.S. regulatory authorities or other applicable United States and non-U.S.
regulatory requirements may, either before or after product approval, if any, subject our company
to administrative or judicially imposed sanctions, including:
| |
|
|
restrictions on the products, manufacturers or manufacturing process; |
| |
|
|
civil and criminal penalties; |
| |
|
|
suspension or withdrawal of regulatory approvals; |
| |
|
|
product seizures, detentions or import bans; |
| |
|
|
voluntary or mandatory product recalls and publicity requirements; |
| |
|
|
total or partial suspension of production; |
| |
|
|
imposition of restrictions on operations, including costly new manufacturing
requirements; and |
| |
|
|
refusal to approve pending NDAs or supplements to approved NDAs. |
Regulatory approval of an NDA or NDA supplement is not guaranteed, and the approval process is
expensive and may take several years. The FDA also has substantial discretion in the drug approval
process. Despite the time and expense exerted, failure can occur at any stage, and we could
encounter problems that cause us to abandon clinical trials or to repeat or perform additional
pre-clinical studies and clinical trials. The number of pre-clinical studies and clinical trials
that will be required for FDA approval varies depending on the drug candidate, the disease or
condition that the drug candidate is designed to address, and the regulations applicable to any
particular drug candidate. The FDA can delay, limit or deny approval of a drug candidate for many
reasons, including:
| |
|
|
a drug candidate may not be deemed safe or effective; |
| |
|
|
the FDA may not find the data from pre-clinical studies and clinical trials sufficient; |
| |
|
|
the FDA might not approve our third-party manufacturers processes or facilities; |
| |
|
|
the FDA may change its approval policies or adopt new regulations; or |
| |
|
|
third party products may enter the market and change approval requirements. |
43
Even if we obtain regulatory approvals for our product candidates, the terms of approvals and
ongoing regulation of our product candidates may limit how we manufacture and market our product
candidates, which could materially impair our ability to generate revenue.
Upon regulatory approval to market any of our product candidates, if any, the approved product and
its manufacturer are subject to continual review. Any regulatory approval that we receive for a
product candidate is likely to be subject to limitations on the indicated uses for which the end product
may be marketed, or include requirements for potentially costly post-approval follow-up clinical
trials. In addition, if the FDA and/or non-U.S. regulatory authorities approve any of our product
candidates, the labeling, packaging, adverse event reporting, storage, advertising and promotion
for the end product will be subject to extensive regulatory requirements. We and the manufacturers
of our products, when and if we have any, will also be required to comply with cGMP regulations,
which include requirements relating to quality control and quality assurance as well as the
corresponding maintenance of records and documentation. Further, regulatory agencies must approve
these manufacturing facilities before they can be used to manufacture our products, when and if we
have any, and these facilities are subject to ongoing regulatory inspection. If we fail to comply
with the regulatory requirements of the FDA and other non-U.S. regulatory authorities, or if
previously unknown problems with our products, when and if we have any, manufacturers or
manufacturing processes are discovered, we could be subject to administrative or judicially imposed
sanctions, including:
| |
|
|
restrictions on the products, manufacturers or manufacturing process; |
| |
| |
|
|
warning letters; |
| |
| |
|
|
civil or criminal penalties or fines; |
| |
| |
|
|
injunctions; |
| |
| |
|
|
product seizures, detentions or import bans; |
| |
| |
|
|
voluntary or mandatory product recalls and publicity requirements; |
| |
| |
|
|
suspension or withdrawal of regulatory approvals; |
| |
| |
|
|
total or partial suspension of production; |
| |
| |
|
|
imposition of restrictions on operations, including costly new
manufacturing requirements; and |
| |
| |
|
|
refusal to approve pending NDAs or supplements to approved NDAs. |
In addition, the FDA and non-U.S. regulatory authorities may change their policies and additional
regulations may be enacted that could prevent or delay regulatory approval of our product
candidates. We cannot predict the likelihood, nature or extent of government regulation that may
arise from future legislation or administrative action, either in the United States, Canada or
abroad. If we are not able to maintain regulatory compliance, we would likely not be permitted to
market our future product candidates and we may not achieve or sustain profitability.
There is a high risk that our drug development activities will not result in commercial products.
Our product candidates are in various stages of development and are prone to the risks of failure
inherent in drug development. We will need to complete significant additional clinical trials
before we can demonstrate that our product candidates are safe and effective to the satisfaction of
the FDA and non-U.S. regulatory authorities. Clinical trials are expensive and uncertain processes
that take years to complete. Failure can occur at any stage of the process, and successful early
clinical trials do not ensure that later clinical trials will be successful. Product candidates in
later-stage clinical trials may fail to show desired efficacy and safety traits despite having
progressed through initial clinical trials. A number of companies in the pharmaceutical industry
have suffered significant setbacks in advanced clinical trials, even after obtaining promising results in earlier clinical trials. In addition, a clinical trial may prove successful
with respect to a secondary objective, but fail to demonstrate clinically significant benefits with
respect to a primary objective. Failure to satisfy a primary objective in a phase 3 clinical trial
(registration trial) would generally mean that a product candidate would not receive regulatory
approval.
44
If government and third-party payors fail to provide coverage and adequate reimbursement rates for
our product candidates, our revenues and potential for profitability will be reduced.
In the United States and elsewhere, our product revenues will depend principally upon the
reimbursement rates established by third-party payors, including government health administration
authorities, managed-care providers, public health insurers, private health insurers and other
organizations. These third-party payors are increasingly challenging the price, and examining the
cost effectiveness, of medical products and services. In addition, significant uncertainty exists
as to the reimbursement status, if any, of newly approved drugs, pharmaceutical products or product
indications. We may need to conduct post-marketing clinical trials in order to demonstrate the
cost-effectiveness of products. Such clinical trials may require us to commit a significant amount
of management time and financial and other resources. If reimbursement of such product is
unavailable or limited in scope or amount or if pricing is set at unsatisfactory levels, our
revenues could be reduced.
In some countries other than the United States, particularly the countries of the European Union
and Canada, the pricing of prescription pharmaceuticals is subject to governmental control. In
these countries, obtaining pricing approval from governmental authorities can take six to twelve
months or longer after the receipt of regulatory marketing approval of a product for an indication.
To obtain reimbursement or pricing approval in some countries, we may be required to conduct a
clinical trial that compares the cost-effectiveness of one of our product candidates to other
available therapies. If reimbursement of such product candidate is unavailable or limited in scope
or amount or if pricing is set at unsatisfactory levels, our revenues could be reduced.
Domestic and foreign governments continue to propose and pass legislation designed to reduce the
cost of healthcare, including drugs. In the United States, there have been, and we expect that
there will continue to be, federal and state proposals to implement similar governmental control.
In addition, increasing emphasis on managed care in the United States will continue to put pressure
on the pricing of pharmaceutical products. For example, the Medicare Prescription Drug Improvement
and Modernization Act of 2003 reforms the way Medicare will cover and reimburse for pharmaceutical
products. The legislation expands Medicare coverage for drug purchases by the elderly and
eventually will introduce a new reimbursement methodology based on average sales prices for certain
drugs. In addition, the new legislation provides authority for limiting the number of outpatient
drugs that will be covered in any therapeutic class. As a result of the new legislation and the
expansion of federal coverage of drug products, we expect that there will be additional pressure to
contain and reduce costs. The Medicaid program and state healthcare laws and regulations may also
be modified to change the scope of covered products and/or reimbursement methodology. Cost control
initiatives could decrease the established reimbursement rates that we receive for any products in
the future, which would limit our revenues and profitability. Legislation and regulations affecting
the pricing of pharmaceutical products, including OGX-011, may change at any time, which could
further limit or eliminate reimbursement rates for OGX-011 or other product candidates.
Failure to obtain regulatory approval outside the United States would prevent us or Teva from
marketing our product candidates abroad.
We intend to market certain of our existing and future product candidates in non-North American
markets. In order to market our existing and future product candidates in the European Union and
many other non-North American jurisdictions, we must obtain separate regulatory approvals. We have
had no interactions with non-North American regulatory authorities, and the approval procedures
vary among countries and can involve additional testing, and the time required to obtain approval
may differ from that required to obtain FDA approval. Approval by the FDA or other regulatory
authorities does not ensure approval by regulatory authorities in other countries, and approval by
one or more non-North American regulatory authorities does not ensure approval by regulatory
authorities in other countries or by the FDA. The non-North American regulatory approval process
may include all of the risks associated with obtaining FDA approval. We may not obtain non-North
American regulatory approvals on a timely basis, if at all. We may not be able to file for non-North American regulatory approvals and may not receive necessary
approvals to commercialize our existing and future product candidates in any market.
45
We are at risk of securities class action litigation.
In the past, securities class action litigation has often been brought against a company following
a decline in the market price of its securities. This risk is especially relevant for us because
our stock price and that of other biotechnology and biopharmaceutical companies have experienced
significant stock price volatility in recent years and particularly over the past year. If we face
such litigation, it could result in substantial costs and a diversion of managements attention and
resources, which could harm our business.
ITEM 1B. UNRESOLVED STAFF COMMENTS
None.
ITEM 2. PROPERTIES
OncoGenex has business offices located in Bothell, Washington and Vancouver, British Columbia.
Prior to the Arrangement, Sonus entered into a non-cancellable lease agreement for laboratory and
office space in Bothell, Washington. Sonus moved into this facility on December 14, 2007. The lease
involves approximately 42,600 square feet of laboratory and office space in a single facility,
currently at a rent of $2 million per annum. The lease has a 10 year term and includes two options
to renew for additional five year periods.
In its Vancouver office, OncoGenex leases approximately 4,857 square feet, currently at a rent of
approximately $181,000 per annum. This lease expires in March 2011. OncoGenex has an option to
renew the lease for a further term of five years.
ITEM 3. LEGAL PROCEEDINGS
From time to time, the Company may be involved in litigation relating to claims arising out of our
operations in the normal course of business. The Company currently is not a party to any legal
proceedings, the adverse outcome of which, in managements opinion, individually or in the
aggregate, would have a material adverse effect on the Companys results of operations or financial
position. There are no material proceedings to which any director, officer or any of our
affiliates, any owner of record or beneficially of more than five percent of any class of our
voting securities, or any associate of any such director, officer, our affiliates, or security
holder, is a party adverse to the Company or its consolidated subsidiary or has a material interest
adverse thereto.
ITEM 4. Reserved
46
PART II
ITEM 5. MARKET FOR THE REGISTRANTS COMMON EQUITY, RELATED STOCKHOLDER MATTERS AND ISSUER
PURCHASES OF EQUITY SECURITIES
Our common stock first began trading on the Nasdaq National Market under the symbol SNUS on
October 12, 1995. Following the completion of the Arrangement discussed elsewhere in this Annual
Report on Form 10-K, our common stock commenced trading on the Nasdaq Capital Market under the
stock symbol OGXI effective August 21, 2008.
No cash dividends have been paid on our common stock, and we do not anticipate paying any cash
dividends in the foreseeable future. As of February 26, 2010, there were approximately 159
shareholders of record and approximately 9,000 beneficial shareholders of our Common Stock. The
high and low sales prices of our common stock as reported by Nasdaq for the periods indicated are
as follows:
| |
|
|
|
|
|
|
|
|
| OncoGenex Pharmaceuticals, Inc |
|
HIGH (1)
|
|
|
LOW (1)
|
|
|
|
|
|
|
|
|
|
|
YEAR ENDED DECEMBER 31, 2008: |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
First quarter |
|
|
10.26 |
|
|
|
6.12 |
|
Second quarter |
|
|
9.00 |
|
|
|
4.50 |
|
Third quarter |
|
|
8.22 |
|
|
|
3.02 |
|
Fourth quarter |
|
|
9.38 |
|
|
|
2.00 |
|
|
|
|
|
|
|
|
|
|
YEAR ENDED DECEMBER 31, 2009: |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
First quarter |
|
|
5.94 |
|
|
|
2.90 |
|
Second quarter |
|
|
27.05 |
|
|
|
3.90 |
|
Third quarter |
|
|
42.99 |
|
|
|
19.30 |
|
Fourth quarter |
|
|
37.88 |
|
|
|
20.10 |
|
| |
|
|
| (1) |
|
All amounts reported herein are presented on a post-one-for-eighteen reverse stock
split basis. |
The information required by this item regarding equity compensation plan information is set forth
in Part III, Item 12 of this Annual Report filed on Form 10-K. We made no purchases of equity
securities during the year ended December 31, 2009.
Recent Sales of Unregistered Securities
On December 24, 2009, pursuant to the Stock Purchase Agreement between the Company and Teva (which
was entered into in connection with the Collaboration Agreement), the Company issued to Teva
267,531 shares of common stock at a price per share of $37.38, for aggregate cash consideration of
approximately $10 million. Such shares have not been registered under the Securities Act of 1933,
as amended (the Securities Act), or any state securities laws, and were issued on a
private placement basis pursuant to the exemptions from registration provided under Section 4(2)
of the Securities Act and Regulation D thereunder, based, in part, on representations by Teva that
it is an accredited investor, as such term is defined in Rule 501 of Regulation D under the
Securities Act. The shares cannot be resold unless they are registered under the Securities Act or
sold pursuant to an exemption therefrom.
47
Stock Performance Graph
The following performance graph shall not be deemed filed for purposes of Section 18 of the
Exchange Act, or incorporated by reference into any filing of the Company under the Securities Act
of 1933, as amended, or the Exchange Act, except as shall be expressly set forth by specific
reference in such filings. The graph compares the cumulative five year total return provided to
shareholders on the Companys common stock relative to the cumulative total returns of the Nasdaq
Composite index and the Nasdaq Pharmaceutical index. An investment of $100 (with reinvestment of all dividends) is assumed to have
been made in our common stock and in each of the indexes on December 31, 2004 and its relative
performance is tracked through December 31, 2009. All amounts reflected in the graph are presented
after having given effect to the one-for-eighteen reverse stock split effected in connection with
the Arrangement.
The stock price performance included in this graph is not necessarily indicative of future stock
price performance.
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
12/04 |
|
|
12/05 |
|
|
12/06 |
|
|
12/07 |
|
|
8/20/08 |
|
|
12/08 |
|
|
12/09 |
|
OncoGenex Pharmaceuticals, Inc. |
|
|
100.00 |
|
|
|
142.49 |
|
|
|
173.09 |
|
|
|
12.32 |
|
|
|
7.93 |
|
|
|
4.72 |
|
|
|
35.06 |
|
NASDAQ Composite |
|
|
100.00 |
|
|
|
101.33 |
|
|
|
114.01 |
|
|
|
123.71 |
|
|
|
106.91 |
|
|
|
73.11 |
|
|
|
105.61 |
|
NASDAQ Pharmaceutical |
|
|
100.00 |
|
|
|
102.23 |
|
|
|
105.16 |
|
|
|
99.56 |
|
|
|
113.74 |
|
|
|
91.99 |
|
|
|
98.21 |
|
| |
|
|
| (1) |
|
August 20, 2008 represents the day before the date of the completion of the
Arrangement. |
48
ITEM 6. SELECTED FINANCIAL DATA
The data set forth below should be read in conjunction with Item 7, Managements Discussion and
Analysis of Financial Condition and Results of Operations and the Financial Statements and Notes
thereto appearing at Item 8 of this report. The selected statements of operations data for the
years ended December 31, 2009, 2008, and 2007 and balance sheet data as of December 31, 2009 and
2008 set forth below have been derived from our audited financial statements included elsewhere in
this Annual Report on Form 10-K. The selected statements of operations data for the years ended
December 31, 2006 and December 31, 2005 and balance sheet data as of December 31, 2007, 2006 and
2005 set forth below have been derived from the audited financial statements for such years not
included in this Annual Report on Form 10-K.
In connection with the Arrangement, OncoGenex Technologies was considered to be the acquiring
company for accounting purposes. Accordingly, the assets and liabilities of Sonus were recorded,
as of the effective time of the Arrangement, at their respective fair values and added to those of
OncoGenex Technologies. The results of the operations and balance sheet data for the year ended
December 31, 2008 reflect the results of only OncoGenex Technologies for the time period of
January 1, 2008 through August 20, 2008 and the results of the combined company from August 21,
2008 through December 31, 2008. The historical results of operations and balance sheet data shown
for years ended December 31, 2007, 2006, and 2005 reflect only those of OncoGenex Technologies
prior to the Arrangement, and do not reflect the results of Sonus. The historical results
presented are not necessarily indicative of future results.
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
December 31, |
|
| |
|
2009 |
|
|
2008 |
|
|
2007 |
|
|
2006 |
|
|
2005 |
|
| |
|
(in thousands except share and per share amounts) |
|
Statements of Operations Data: |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Collaboration revenue |
|
$ |
25,539 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Operating expenses |
|
$ |
28,121 |
|
|
$ |
11,112 |
|
|
$ |
7,675 |
|
|
$ |
11,302 |
|
|
$ |
4,666 |
|
Net loss |
|
$ |
5,476 |
|
|
$ |
4,204 |
|
|
$ |
8,536 |
|
|
$ |
11,594 |
|
|
$ |
4,929 |
|
Redeemable
convertible
preferred share
accretion |
|
$ |
|
|
|
$ |
1,973 |
|
|
$ |
2,944 |
|
|
$ |
2,604 |
|
|
$ |
1,843 |
|
Loss attributable to
common shareholders |
|
$ |
5,476 |
|
|
$ |
6,177 |
|
|
$ |
11,480 |
|
|
$ |
14,198 |
|
|
$ |
6,772 |
|
Basic and diluted
loss per common
share |
|
$ |
(0.95 |
) |
|
$ |
(3.38 |
) |
|
$ |
(96.63 |
) |
|
$ |
(119.51 |
) |
|
$ |
(58.71 |
) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Shares used in calculation of net loss
per share |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Basic |
|
|
5,766,850 |
|
|
|
1,829,276 |
|
|
|
118,801 |
|
|
|
118,801 |
|
|
|
115,350 |
|
Diluted |
|
|
5,766,850 |
|
|
|
1,829,276 |
|
|
|
118,801 |
|
|
|
118,801 |
|
|
|
115,350 |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
December 31, |
|
| |
|
2009 |
|
|
2008 |
|
|
2007 |
|
|
2006 |
|
|
2005 |
|
| |
|
(in thousands) |
|
Balance Sheet Data: |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash, cash equivalents
and marketable
securities |
|
$ |
64,568 |
|
|
$ |
12,419 |
|
|
$ |
5,131 |
|
|
$ |
8,012 |
|
|
$ |
13,785 |
|
Total assets |
|
$ |
68,980 |
|
|
$ |
14,790 |
|
|
$ |
7,350 |
|
|
$ |
9,395 |
|
|
$ |
19,750 |
|
Current liabilities |
|
$ |
25,781 |
|
|
$ |
2,884 |
|
|
$ |
8,200 |
|
|
$ |
2,532 |
|
|
$ |
1,752 |
|
Series preferred shares |
|
$ |
|
|
|
$ |
|
|
|
$ |
37,373 |
|
|
$ |
34,429 |
|
|
$ |
31,825 |
|
Common shares |
|
$ |
73,804 |
|
|
$ |
56,076 |
|
|
$ |
399 |
|
|
$ |
399 |
|
|
$ |
399 |
|
Accumulated deficit |
|
$ |
(53,485 |
) |
|
$ |
(48,009 |
) |
|
$ |
(41,832 |
) |
|
$ |
(30,352 |
) |
|
$ |
(16,154 |
) |
Shareholders equity |
|
|
22,959 |
|
|
|
10,707 |
|
|
|
(38,223 |
) |
|
|
(27,566 |
) |
|
|
(13,827 |
) |
49
ITEM 7. MANAGEMENTS DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF OPERATIONS
Forward-Looking Statements
This document contains forward-looking statements within the meaning of the Private Securities
Litigation Reform Act of 1995. These forward-looking statements involve a number of risks and
uncertainties. We caution readers that any forward looking statement is not a guarantee of future
performance and that actual results could differ materially from those contained in the
forward-looking statement. These statements are based on current expectations of future events.
Such statements include, but are not limited to, statements about future financial and operating
results, plans, objectives, expectations and intentions, costs and expenses, interest rates,
outcome of contingencies, financial condition, results of operations, liquidity, business
strategies, cost savings, objectives of management and other statements that are not historical
facts. You can find many of these statements by looking for words like believes, expects,
anticipates, estimates, may, should, will, could, plan, intend, or similar
expressions in this document or in documents incorporated by reference in this document. We intend
that such forward-looking statements be subject to the safe harbors created thereby. Examples of
these forward-looking statements include, but are not limited to:
| |
|
|
our anticipated future capital requirements and the terms of any capital financing
agreements; |
| |
|
|
progress and preliminary and future results of clinical trial conducted by us or our
collaborators; |
| |
|
|
anticipated regulatory filings, requirements and future clinical trials conducted by
us or our partners; |
| |
|
|
timing and amount of future contractual payments, product revenue and operating
expenses; and |
| |
|
|
market acceptance of our products and the estimated potential size of these markets. |
These forward-looking statements are based on the current beliefs and expectations of our
management and are subject to significant risks and uncertainties. If underlying assumptions prove
inaccurate or unknown risks or uncertainties materialize, actual results may differ materially
from current expectations and projections. The following factors, among others, could cause actual
results to differ from those set forth in the forward-looking statements:
| |
|
|
uncertainties regarding our future operating results, and the risk that our product
candidates will not obtain the requisite regulatory approvals to commercialize or that
the future sales of our product candidates may be less than expected or nil; |
| |
|
|
future capital requirements and uncertainty of obtaining additional funding through
debt or equity financings on terms acceptable to us; |
| |
|
|
dependence on Tevas ongoing commitment and ability to develop and commercialize
OGX-011; |
| |
|
|
dependence on the development and commercialization of our product candidates,
particularly on OGX-011; |
| |
|
|
the risk that previous clinical trial results may not be indicative of results in
future studies; |
| |
|
|
the risk that results of research and preclinical studies may not be indicative of
results in humans; |
| |
|
|
uncertainty relating to the timing and results of clinical trials; |
| |
|
|
uncertainties regarding the safety and effectiveness of our products and technologies; |
| |
|
|
the timing, expense and uncertainty associated with the development and regulatory
approval process for products; |
| |
|
|
acceptance of our products by the medical community; |
| |
|
|
the uncertainty associated with exiting or subleasing our excess office and laboratory
space; |
| |
|
|
our ability to build out our product candidate pipeline through product in-licensing
or acquisition activities; |
| |
|
|
our future dependence on Teva to market and promote OGX-011 and to provide us with
accurate financial data; |
| |
|
|
general competitive conditions within the drug development and pharmaceutical industry
and new
development s or therapies that may not work in combination with our product candidates; |
| |
|
|
our dependence on key employees; |
50
| |
|
|
proper management of our operations will be critical to the success of the Company; |
| |
|
|
the potential for product liability issues and related litigation; |
| |
|
|
the potential for claims arising from the use of hazardous materials in our business; |
| |
|
|
the potential inability to successfully protect and enforce our intellectual property
rights; |
| |
|
|
the reliance on third parties who license intellectual property rights to us to comply
with the terms of such agreements and to enforce, prosecute and defend such intellectual
property rights; |
| |
|
|
the impact of current, pending or future legislation, regulations and legal actions in
the United States, Canada and elsewhere affecting the pharmaceutical and healthcare
industries; |
| |
|
|
volatility in the value of our common stock; and |
| |
|
|
general economic conditions. |
You are cautioned not to place undue reliance on these forward-looking statements, which speak
only as of the date of this document or, in the case of documents referred to or incorporated by
reference, the date of those documents.
All subsequent written or oral forward-looking statements attributable to us or any person acting
on our behalf are expressly qualified in their entirety by the cautionary statements contained or
referred to in this section. We do not undertake any obligation to release publicly any revisions
to these forward-looking statements to reflect events or circumstances after the date of this
document or to reflect the occurrence of unanticipated events, except as may be required under
applicable U.S. securities law. If we do update one or more forward-looking statements, no
inference should be drawn that we will make additional updates
with respect to those or other forward-looking statements.
MD&A Overview
In this Managements Discussion and Analysis of Financial Condition and Results of Operations we
explain the general financial condition and the results of operations for our Company, including:
| |
|
|
an overview of our business; |
| |
|
|
results of operations and why those results are different from the prior year; and |
| |
|
|
capital resources we currently have and possible sources of additional funding for
future capital requirements. |
Arrangement Agreement
As discussed in the notes to the financial statements below, during 2008, we completed the
Arrangement with OncoGenex Technologies and, in connection therewith, effected a one-for-eighteen
reverse stock split. All information in this report relating to the number of shares, price per
share, and per share amounts of common stock are presented on a post-reverse stock split basis. For
more information concerning the Arrangement, see the discussion of the Arrangement in note 5 to the
financial statements included in this Annual Report on Form 10-K.
Overview of the Company and 2009 Developments
OncoGenex is a biopharmaceutical company committed to the development and commercialization of new
cancer therapies that address unmet needs in the treatment of cancer. We have five product
candidates in our pipeline, with each product candidate having a distinct mechanism of action and
representing a unique opportunity for cancer drug development.
51
Our product candidates OGX-011, OGX-427 and OGX-225 focus on mechanisms of treatment resistance in
cancer patients and are designed to address treatment resistance by blocking the production of
specific proteins which we believe promote survival of tumor cells and are over-produced in
response to a variety of cancer treatments. Our aim in targeting these particular proteins is to
disable the tumor cells adaptive defenses and thereby render the tumor cells more susceptible to
attack with a variety of cancer therapies, including chemotherapy, which we believe will increase
survival time and improve the quality of life for cancer patients. Product candidate SN2310 is a
novel camptothecin for the treatment of cancer. Camptothecins are potent anticancer agents that
belong to the family of drugs called topoisomerase I inhibitors that bind reversibly to the
TOPO-I-DNA complex causing breaks in the DNA strands during replication resulting in cell death.
Product candidate CSP-9222 is the lead compound from a family of compounds, which have been
in-licensed from Bayer, that demonstrate activation of programmed cell death in pre-clinical
models.
Product Candidate OGX-011
As discussed above, in December 2009, we announced our entry into the Collaboration Agreement with
Teva, for the development and global commercialization of OGX-011 (and related compounds targeting
clusterin with the exclusion of OGX-427 and OGX-225).
OncoGenex and Teva have developed a Clinical Development Plan under which three phase 3 clinical
trials will be initiated. We have designed two of the phase 3 clinical trials to evaluate the
clinical benefit of OGX- 011 in CRPC and, together with Teva, we are in the process of designing a
third phase 3 clinical trial evaluating the clinical benefit of OGX-011 in NSCLC, as follows:
| |
|
|
Evaluating a survival benefit for OGX-011 in combination with first-line docetaxel
treatment in approximately 800 men with CRPC; |
| |
|
|
Evaluating a durable pain palliation benefit for OGX-011 in combination with docetaxel
as second-line chemotherapy in approximately 300 men with CRPC; and |
| |
|
|
Evaluating a survival benefit for OGX-011 in combination with first-line chemotherapy
in at least 700 patients with NSCLC. |
For detailed information regarding our relationship with Teva and the Collaboration Agreement,
refer to the discussion under the heading License and Collaboration Agreements Teva
Pharmaceutical Industries Ltd., which is incorporated herein by reference.
During 2009, OGX-011 received Fast Track designations from the FDA for the treatment of progressive
metastatic prostate cancer in combination with docetaxel for both first and second-line docetaxel
treatment. The FDA has agreed on the design of two phase 3 registration trials, each in CRPC, via
the SPA process. One trial design investigates overall survival as the primary endpoint for OGX-011
in combination with first-line chemotherapy, whereas the other trial design investigates pain
palliation as the primary endpoint for OGX-011 in combination with second-line chemotherapy.
As discussed above, in February of 2010, OGX-011 received written, scientific advice from the EMA
on the Companys development plan for OGX-011 for the treatment of men with CRPC. The input
received from the CHMP at the EMA was in overall agreement with OncoGenexs development plan
regarding the proposed preclinical studies and both the study designs and analyses for the phase
III trials. The CHMP also agreed that the intended safety database would enable a sufficiently
qualified risk-benefit assessment for market approval.
Final results of a randomized phase 2 trial evaluating the benefit of combining OGX-011 with
first-line docetaxel chemotherapy were presented during an oral presentation at the ASCO 2009
Annual Meeting. Analyses indicating a survival benefit in patients treated with OGX-011 in
combination with first-line docetaxel compared to docetaxel alone, the latter of which being the
current standard care for patients with
advanced, progressive metastatic prostate cancer, are described above under the heading Summary of
Final Results of OGX-011 Phase 2 Clinical Trial in First-Line Hormone Refractory Prostate Cancer,
which is incorporated herein by reference.
52
Due to the final results of this randomized phase 2 trial, the phase 3 registration trial that
Teva will initiate in 2010 will evaluate the overall survival benefit of OGX-011 in patients
treated with first-line chemotherapy.
Durable pain palliation defined as pain palliation of 12 weeks or greater has been observed in
another phase 2 trial evaluating patients with metastatic CRPC who progressed while receiving, or
within 6 months of completing, first-line docetaxel treatment. In this trial, 46% of patients who
were retreated with docetaxel as second-line treatment in combination with OGX-011 had durable pain
palliation. This is favorable even when compared to the 35% pain responses of 3 weeks or greater
observed in the phase 3 study which supported the registration of docetaxel as first-line
chemotherapy in patients with CRPC. Due to the results of our phase 2 trial, the phase 3
registration trial that OncoGenex will initiate in 2010 will evaluate the durable pain palliation
benefit of OGX-011 in patients treated with second-line chemotherapy.
Product Candidate OGX-427
A phase 1 trial has evaluated 53 patients with a variety of cancers, with enrollment ongoing.
OGX-427 was first evaluated as a single agent in a dose escalation manner up to 1000mg OGX-427. A
maximum tolerated dose was not identified up to and including the 1000mg dose of OGX-427
monotherapy. Subsequently, as defined by the protocol, an 800mg dose of OGX-427 in combination with
docetaxel was evaluated, followed by a 1000mg OGX-427 plus docetaxel. OGX-427 is administered as
three loading doses within the first nine days and then continued weekly, with three weeks defined
as a treatment cycle, until disease progression or toxicity. In those groups receiving OGX-427 in
combination with docetaxel, 75mg/M2 docetaxel was administered on day 1 of every 3-week cycle
starting after completion of the OGX-427 loading doses.
Preliminary results of this phase 1 trial were presented during an oral presentation at the ASCO
2009 Annual Meeting. At that time, 41 patients were enrolled who had a diagnosis of breast,
ovarian, prostate or non-small cell lung cancer and most had failed multiple prior chemotherapy
treatments. A median of two cycles (range of one to eight cycles) was administered.
OGX-427 treatment was well tolerated as a monotherapy. No evidence of altered cardiac activity was
observed. A majority of adverse events were mild and mainly occurred during the loading doses.
Adverse events consisted of chills, itching and fatigue in over one-third of patients. There was a
trend for increasing incidence of some mild adverse events with escalating OGX-427 doses. For
example, 33% of patients at the 200mg dose compared to 67% of patients at the 1000mg dose had mild
adverse events during the loading doses. The half-life of OGX-427 in the blood remained constant,
although there appeared to be an increase in maximum blood levels and a corresponding decrease in
blood clearance of OGX-427 as doses were escalated.
The combination of OGX-427 with docetaxel at both dose levels was also well tolerated. This data is
subject to further analysis.
CTCs, an emerging metric to assess treatment effect, were evaluated at baseline before treatment
and during OGX-427 treatment as a monotherapy. Both total and Hsp27-positive CTCs were evaluated.
Declines of 50% or greater in both total and Hsp27-positive CTCs were observed in over one-half of
the patients in each cohort and in each type of cancer. Declines in Hsp27 CTCs to 5 or less cells
occurred in 27% of patients who had greater than 5 CTCs at baseline. Reduction in tumor markers
defined as declines of prostate specific antigen, or PSA, levels in prostate cancer or CA-125
levels in ovarian cancer were also observed. A reduction in PSA level was observed in 7 of 20
patients (35%) with prostate cancer and a reduction in CA-125 levels was observed in 3 of 5
patients (60%) with ovarian cancer.
53
A second investigator-sponsored phase 1 clinical trial evaluating OGX-427 when administered
directly into the bladder in patients with bladder cancer was initiated in August 2009. The study,
which will enroll up to 36 patients with bladder cancer, is designed to determine the safety and
potential benefit of OGX-427 administered directly into the bladder using a catheter, which is
known as intravesical instillation. In addition, the study will measure the direct effect of
OGX-427 on expression of Hsp27 in bladder tumor cells as well as determine the pharmacokinetics and
pharmacodynamics of OGX-427 when delivered by
intravesical instillation. This investigator-sponsored study is funded by the National Cancer
Institute of Canada (NCIC).
In January 2010, we announced that an investigator-sponsored phase 2 clinical trial evaluating
OGX-427 when administered as a monotherapy to patients with CRPC has received grant funding. The
randomized, controlled phase 2 study will enroll up to 72 patients and is designed to determine the
potential benefit of OGX-427 by evaluating the number of patients who are without disease
progression at 12 weeks post study treatment with or without OGX-427. This phase 2 trial will also
measure the direct effect of OGX-427 on PSA levels, time to progression by PSA or measurable
disease, numbers of CTCs and other relevant secondary endpoints. The trial is expected to start by
mid 2010 following final analysis of phase 1 data and approval by Health Canada and the
institutional review board. The funds were awarded by a third party granting agency to Dr. Kim Chi,
a medical oncologist at the BC Cancer Agency, Research Scientist at the Vancouver Prostate Centre
and the principal investigator of the OGX-427 phase 2 trial.
Product Candidates OGX-225, SN2310 and CSP-9222
SN2310 was evaluated in a phase 1 clinical trial to evaluate safety in patients with advanced
cancer who have received on average three to five prior chemotherapy treatments. SN2310 has been
administered to 26 patients with various types of cancer in a phase 1 clinical trial. The phase 1
clinical trial has been completed and the dose-limiting toxicity that defined a maximum tolerated
dose in this heavily pretreated patient population has been determined. We do not intend to
initiate additional trials for SN2310 but rather will seek to out-license or sell this product
candidate to a third party. OGX-225, an inhibitor of insulin growth factor binding proteins 2 and
5, and CSP-9222 are in pre-clinical development.
Collaboration Revenue
We recorded $25.5 million of collaboration revenue in connection with our OGX-011 Collaboration
Agreement with Teva in the year ended December 31, 2009. At December 31, 2009, $26.5 million of
the upfront payment was included in the balance sheet line item Deferred Collaboration Revenue,
which we are amortizing over a period of approximately 3 years based on the expected performance
period of our deliverables under this agreement. Further, we are eligible to receive payments of
up to $370 upon the achievement of developmental and commercial milestones. At present, we are
unable to predict the timing or likelihood of such milestone payments, although we do not expect
to receive any milestone payments from Teva in the year ended December 31, 2010. There were no
revenues in 2008 or 2007. See note 4 in the Notes to Financial Statements for further details on
our collaboration with Teva.
Research and Development Expenses
Research and development (R&D) expenses consist primarily of costs for: milestone payments to
third parties, clinical trials; materials and supplies; facilities; personnel, including salaries
and benefits; regulatory activities; pre-clinical studies; licensing and intellectual property; and
allocations of other research and development-related costs. External research and development
expenses include fees paid to universities, hospitals and other entities that conduct certain
research and development activities and that manufacture our product candidates for use in our
clinical trials. We expect our research and development expenses to increase significantly in the
future as we continue to develop our product candidates. Currently, OncoGenex manages its clinical
trials through independent medical investigators at their sites and at hospitals.
Under the Collaboration Agreement with Teva, we are required to spend $30 million towards
development of OGX-011 which will include personnel costs for certain development activities. Teva
will fund all other expenses under the Clinical Development Plan. A total of $3.5 million of costs incurred by the
Company in 2009 were applied against the Companys $30 million funding commitment, resulting in a
remaining funding commitment of $26.5 million at December 31, 2009. We expect full time equivalent
reimbursement of between $1.5 and $2.5 million per year from 2010 to 2012. We expect to incur the
remaining costs associated with the Clinical Development Plan over the next three years.
54
A majority of the Companys expenditures to date have been related to the development of OGX-011.
Until July 2, 2008, OGX-011 was being co-developed with Isis and R&D expenses for OGX-011 were
shared on the basis of 65% OncoGenex and 35% Isis. On July 2, 2008, OncoGenex and Isis amended
their agreement to provide for unilateral development of OGX-011 by OncoGenex. In connection with
the Collaboration Agreement and pursuant to the terms of agreements between the Company and Isis
relating to OGX-011, the Company accrued a payment of $10 million to Isis, which was included in
R&D expenses in the period. The Company also accrued a payment of approximately $333,333 to UBC
pursuant to the terms of their license agreement relating to OGX-011, which was also included in
R&D expenses in the period.
Several of the Companys clinical trials have been supported by grant funding which was received
directly by the hospitals and/or clinical investigators conducting the clinical trials allowing
OncoGenex to complete these clinical trials with minimal expense.
Since the Companys drug candidates are in the early stage of development, we cannot estimate
completion dates for development activities or when we might receive material net cash inflows from
our research and development projects.
We expect our research and development expenses to increase in 2010 as we further expand
development of OGX-011, OGX-427 and our other programs. Our projects or intended projects may be
subject to change from time to time as we evaluate our research and development priorities and
available resources.
General and Administrative Expenses
General and administrative (G&A) expenses consist primarily of salaries and related costs for
our personnel in executive, business development, human resources, external communications,
finance and other administrative functions, as well as consulting costs, including market research
and business consulting. Other costs include professional fees for legal and auditing services,
insurance and facility costs. We believe that G&A resources are sufficient to carry on existing
development activities. We anticipate that G&A expenses will increase significantly in the future
as we continue to expand our operating activities.
Restructuring Activities
On August 21, 2008, immediately following the completion of the Arrangement, the Company reduced
its workforce by approximately 49%. Severance payable at the date of the restructuring in
connection with former employees of Sonus was $1,322,000 and has been accounted for in accordance
with EITF No. 95-3, Recognition of Liabilities in Connection with a Purchase Business Combination
as part of the purchase price allocation. During 2008, the Company made payments totalling
$1,186,000 and the amount owing at December 31, 2008 was $137,000. All remaining severance
liabilities relating to transaction-related workforce reductions were paid out during 2009, and the
amount owing at December 31, 2009 was nil.
Prior to the Arrangement, Sonus entered into a non-cancellable lease arrangement for office space
located in Bothell, Washington, which is considered to be in excess of the Companys current
requirements. The Company is currently in the process of evaluating opportunities to exit or sublet
portions of the leased space and recorded an initial restructuring charge of $2,084,000 on August
21, 2008 as part of the purchase price allocation. The liability is computed as the present value
of the difference between the remaining lease payments due less the estimate of net sublease income
and expenses and has been accounted for in accordance with EITF No. 95-3, Recognition of
Liabilities in Connection with a Purchase Business Combination. This represents the Companys best
estimate of the fair value of the liability. Subsequent changes in the liability due to accretion, or changes in estimates of sublease assumptions will be
recognized as adjustments to restructuring charges in future periods. During 2008, $362,000 was
amortized into income, resulting in a remaining liability.
55
In June 2009 the Company revised its sublease income assumptions used to estimate the fair value of
the excess lease facility liability. These assumptions were subsequently revised again in December
2009. These changes in estimate resulted in increases in the fair value of the excess lease
liability and $494,000 and $3,547,000 in charges to research and development expense recorded in
June 2009 and December 2009, respectively, to reflect these changes in estimate. These changes in
estimate had a $0.69 impact on loss per common share for year ended December 31, 2009. The
estimated carrying value of the liability remaining at December 31, 2009 with respect to excess
facilities is $4,645,000.
Results of Operations
The consolidated financial statements reflect the Arrangement between Sonus and OncoGenex
Technologies as a reverse acquisition, whereby OncoGenex Technologies is deemed to be the
acquiring entity from an accounting perspective. Accordingly, for the year ended December 31,
2008, the consolidated results of operations of the Company include only the results of operations
of OncoGenex Technologies for the time period of January 1, 2008 through August 21, 2008 and the
results of the combined company following the completion of the Arrangement on August 21, 2008.
The consolidated results of operations for the years ended December 31, 2007 include only the
consolidated results of operations of OncoGenex Technologies and do not include historical results
of Sonus. This treatment and presentation is in accordance with SFAS 141, Business Combinations.
Proforma results are included in note 5 to the financial statements.
Years Ended December 31, 2009 and December 31, 2008
R&D expenses for the year ended December 31, 2009 increased to $24.2 million from $7.8 million for
the year ended December 31, 2008, due mainly to higher OGX-011 and OGX-427 development costs,
milestone payments owed to Isis and UBC resulting from the Collaboration Agreement with Teva, an
increase in employee expenses and higher facility costs resulting from the Arrangement. Included
in the year ended December 31, 2008 was a SRED claim of $0.6 million, which offset R&D expenses in
the period. Since OncoGenex Technologies ceased to be a Canadian Controlled Private Company under
Canadian tax laws as a result of the Arrangement, SRED claims can now only be applied against
taxes payable. The SRED program is a Canadian federal tax incentive program that encourages
Canadian businesses to conduct research and development in Canada.
G&A expenses for the year ended December 31, 2009 increased to $4.0 million from $3.3 million for
the year ended December 31, 2008, due mainly to higher employee expenses and increased costs
associated with operating as a public company.
Interest income for the year ended December 31, 2009 decreased to $47 thousand from $210 thousand
for year ended December 31, 2008. Of the $210 thousand in interest for the 2008 period, $60
thousand related to interest received from the Canada Revenue Agency in relation to the Companys
2006 Scientific Research and Development claim, while the 2009 amount includes only interest
earned on cash and cash equivalents and marketable securities.
Other for the year ended December 31, 2009 decreased to $70 thousand in income from to $211
thousand in income for the year ended December 31, 2008, due to lower gains on sales of equipment.
Years Ended December 31, 2008 and December 31, 2007
R&D expenses for the year ended December 31, 2008 were $7.8 million compared to $4.1 million for
the year ended December 31, 2007, which reflects an increase of $3.7 million due mainly to costs
associated with the development of OGX-427, an increase in employee expenses and higher facility
costs both resulting from the reverse takeover of Sonus.
56
G&A expenses for the year ended December 31, 2008 were $3.3 million compared to $3.5 million for
the year ended December 31, 2007, which reflects a decrease of approximately $200 thousand due
mainly to higher costs associated with employee expenses and increased costs associated with
operating as a public company, offset by higher costs incurred as part of a planned initial public
offering in 2007 by OncoGenex Technologies which was later abandoned.
Interest income for the year ended December 31, 2008 was $210 thousand compared to $177 thousand
for year ended December 31, 2007, which reflects an increase of $33 thousand due mainly to an
increase in cash equivalents and short term investments.
Other for the year ended December 31, 2008 was $211 thousand in income compared to $325 thousand
in expense for the year ended December 31, 2007, due to gains on sales of equipment, and foreign
exchange gains.
Liquidity and Capital Resources
OncoGenex has incurred an accumulated deficit of $53.5 million through December 31, 2009, and we
expect to incur substantial and increasing additional losses in the future as we expand our
research and development activities. We have not generated any revenue from product sales to date,
and we do not expect to generate product sales revenue for several years, if ever. In the year
ended December 31, 2009, we generated $25.5 million in collaboration revenue from the Teva
Collaboration Agreement.
All of our operations to date have been funded through the sale of our debt and equity securities,
and upfront payments received from Teva. As at December 31, 2009, OncoGenex had cash, cash
equivalents, and short-term investments of $64.6 million in the aggregate as compared to cash,
cash equivalents and short-term investments of $12.4 million as at December 31, 2008. As at
December 31, 2009, OncoGenex does not have any borrowing or credit facilities available to it.
Based upon our current expectations, we believe our capital resources at December 31, 2009 will be
sufficient to fund our currently planned operations into 2012 and
expect that both phase 3 prostate cancer trials will be fully accrued
by this time. We may seek additional funding through, among other things, executing a partnership or
collaboration agreement, or licensing or sale of certain of our product
candidates, or through private or public offerings of our equity securities or debt financings.
Our currently planned operations
are set forth below under the heading Operating Capital and Capital Expenditure Requirements.
Cash Flows
Cash Used in Operations
For the years ended December 31, 2009, net cash provided by operating activities was
$34.9 million, compared to $12.3 million of net cash used in operation in 2008. This increase in
cash provided by operating activities during 2009 was driven by the upfront cash payment from
Teva, offset by increased R&D expenses associated with personnel and facilities assumed in the
Arrangement, cash used to reduce liabilities assumed in the Arrangement and increased current
assets associated with R&D activities.
For the year ended December 31, 2008, cash used in operations of $7.9 million was attributable
primarily to our losses from operations, offset by an increase in investment tax credit
recoverable of $1.0 million, and an increase in taxes payable under Part VI.1 of the Income Tax
Act (Part VI.1 tax) of $1.0 million and the interest on its convertible debentures of $0.2
million.
Cash Provided by Financing Activities
For the years ended December 31, 2009 and 2008, net cash provided by financing activities was
$17.3 million and $121 thousand respectively. Net cash provided by financing activities in the
year ended December 31, 2009 was attributable to the net proceeds we received from the issuance of
common shares through a registered direct offering, net proceeds we received from the issuance of
common shares to Teva as part of the Share Purchase Agreement, and the proceeds from the issuance
of common shares on stock option exercises. All net cash provided by financing activities in the
year ended December 31, 2008 was the result of proceeds from the issuance of common shares on stock option exercises, offset by cash
paid on the elimination of fractional shares following the one-for-eighteen reverse stock split.
All net cash provided by financing activities in the year ended December 31, 2007 was due to the
issuance of convertible debentures.
57
Cash Used/Provided by Investing Activities
Net cash provided by investing activities for the year ended December 31, 2009 and December 31,
2008 was $2.2 million and $15.1 million, respectively. Net cash provided by investing activities
in the year ended December 31, 2009 was due to transactions involving marketable securities in the
normal course of business. Net cash provided by investing activities in the year ended December
31, 2008 was due to the Arrangement with Sonus and transactions involving marketable securities in
the normal course of business.
Net cash provided by investing activities for the years ended December 31, 2007 of $6.3 million
was due primarily to maturities of investments.
Operating Capital and Capital Expenditure Requirements
We believe that our cash, cash equivalents and short-term investments will be sufficient to fund
our currently planned operations into 2012, including:
| |
|
|
completing patient accrual in a phase 3 clinical trial
evaluating a durable pain palliation benefit for OGX-011 in
combination with docetaxel as second-line chemotherapy in approximately 300 men with CRPC
which we expect to initiate in the second quarter of 2010; |
| |
|
|
completing patient accrual in a phase 3 clinical trial evaluating a survival benefit for
OGX-011 in combination with docetaxel as first-line chemotherapy in approximately
800 men with CRPC which we expect to initiate in the third quarter of 2010; |
| |
|
|
completing follow-up monitoring visits related to our completed phase 2 clinical
trials of OGX-011; |
| |
|
|
completing follow-up monitoring visits related to the phase 1 clinical trial
evaluating OGX-427 as a monotherapy in patients with solid tumors and continuing
evaluation of OGX-427 in combination with docetaxel in patients with solid tumors; |
| |
|
|
continuing an investigator-sponsored phase 1 clinical trial evaluating OGX-427
treatment in patient with bladder cancer; |
| |
|
|
initiating an investigator-sponsored phase 3 clinical trial evaluating OGX-427
treatment in patients with prostate cancer; and |
| |
|
|
meeting working capital needs, capital expenditures and general corporate purposes. |
As of December 31, 2009 we have a remaining commitment to fund $26.5 million towards the
three phase 3 trials of OGX-011, while Teva is required to fund
all additional expenses under the clinical development plan. The final results from
the phase 3 trials may be released at a date beyond our current available cash runway.
In addition, if we desire to conduct development activities with respect to our other
product candidates beyond those development activities mentioned in the list above,
we will require additional funding to support such operations. If and when needed to extend our cash availability
or to conduct any such currently unplanned development activities, we would seek any such necessary funding through
the licensing or sale of certain of our product candidates, executing a partnership or collaboration agreement,
or through private or public offerings of our equity securities or debt financings.
Our future capital requirements will depend on many factors including:
| |
|
|
maintaining Teva relationship and Tevas ongoing level of focus and efforts to develop
OGX-011; |
| |
|
|
our ability to obtain additional funding through a partnership or collaboration
agreement with a third party or licenses of certain of our product candidates, or through
private or public offerings of our equity securities or debt financings; |
| |
|
|
timing and costs of clinical trials, preclinical development and regulatory approvals; |
| |
|
|
timing and cost of drug discovery and research and development; |
| |
|
|
entering into new collaborative or product license agreements for products in our
pipeline; and |
| |
|
|
costs related to obtaining, defending and enforcing patents. |
58
Contractual Obligations
The following table summarizes our contractual obligations as of December 31, 2009:
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
Less than |
|
|
|
|
|
|
|
|
|
|
More than |
|
| Contractual Obligations |
|
Total |
|
|
1 year |
|
|
1-3 years |
|
|
3-5 years |
|
|
5 years |
|
Bothell office operating lease (1) |
|
$ |
17,743,000 |
|
|
$ |
1,995,000 |
|
|
$ |
4,172,000 |
|
|
$ |
4,426,000 |
|
|
$ |
7,150,000 |
|
Vancouver office operating lease
(2) |
|
$ |
226,000 |
|
|
$ |
181,000 |
|
|
$ |
45,000 |
|
|
|
|
|
|
|
|
|
Bayer license maintenance fees (3) |
|
$ |
875,000 |
|
|
$ |
125,000 |
|
|
$ |
325,000 |
|
|
$ |
425,000 |
|
|
|
|
|
UBC license maintenance fees (4) |
|
$ |
38,200 |
|
|
$ |
7,600 |
|
|
$ |
15,300 |
|
|
$ |
15,300 |
|
|
|
|
|
Leased equipment |
|
$ |
67,300 |
|
|
$ |
41,800 |
|
|
$ |
20,200 |
|
|
|
5,300 |
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total |
|
$ |
18,949,500 |
|
|
$ |
2,350,400 |
|
|
$ |
4,577,500 |
|
|
$ |
4,871,600 |
|
|
$ |
7,150,000 |
|
| |
|
|
| (1) |
|
This operating lease, which commenced in 2007, is for a term of ten years and
contains a provision for two additional five-year renewals. |
| |
| (2) |
|
This operating lease expires in 2011. |
| |
| (3) |
|
Under the terms of our agreement with Bayer, OncoGenex will make annual payments to
Bayer on June 27 of each year (Anniversary Payments), with an initial payment of $100,000 in
2008. The payments will increase annually by $25,000 until the initiation of the first phase 3
clinical trial related relating to CSP-9222, at which point the Anniversary Payments reset to
$100,000 and increase by $25,000, until such time as the Company achieves either the first NDA
filing in the United States or the European Union related to CSP-9222. For the purposes of this
table we assume no reset in pricing resulting from initiation of phase 3 trials. OncoGenex has the
option to terminate this contract upon 60 days written notice to Bayer. |
| |
| (4) |
|
The Company is obligated to pay an annual license maintenance fee of CAD $8,000 to
UBC, which has been translated based on the December 31, 2009 exchange rate of US$1.00 =
CAD$1.224, and rounded to the nearest $100. |
Off-Balance Sheet Arrangements
We do not have any off-balance sheet financing arrangements at December 31, 2009.
Inflation
We not believe that inflation has had a material impact on our business and operating results
during the periods presented.
Contingencies and Commitments
Teva Pharmaceutical Industries Ltd.
Under the Collaboration Agreement, the Company is required to contribute $30 million in direct and
indirect costs towards the Clinical Development Plan. $3.5 million of these costs were incurred by
OncoGenex during 2009, resulting in a remaining funding responsibility of $26.5 million which has
been recorded on the Companys balance sheet as Deferred Collaboration Revenue. Teva will fund all
other expenses under the Clinical Development Plan.
In addition to the development costs noted above, Teva is also responsible for all costs relating
to product commercialization including costs incurred in relation to the Companys co-promotion
option, except for start-up costs in advance of commercialization.
59
Isis Pharmaceuticals Inc. and University of British Columbia
Pursuant to license agreements the Company has with the UBC and Isis, the Company is obligated to
pay royalties on future product sales and milestone payments of up to
$10.1 million upon the
achievement of specified product development milestones related to OGX-427 and OGX-225.
In addition, we are required to pay to Isis 30% of all Non-Royalty Revenue we receive. Isis has
disclosed in its SEC filings that it is entitled to receive 30% of the up to $370 million in
milestone payments we may receive from Teva as part of the Collaboration Agreement; however, we
believe that certain of the milestone payments related to sales targets may qualify as Royalty
Revenue, and therefore be subject to the lesser payment obligations. No
assurance can be provided that we will be entitled to receive these milestone payments or, if we
are, that the applicable amount payable to Isis will be less than 30%.
We are also obligated to pay to UBC certain patent costs and annual license maintenance fees for
the extent of the patent life of CAD $8,000 per year. We anticipate paying Isis $750,000 in 2010
upon the initiation of a phase 2 clinical trial of OGX-427 in patients with CRPC. We do not
anticipate making any royalty payments to Isis in 2010.
The UBC agreements have effective dates ranging from November 1, 2001 to April 5, 2005 and each
agreement expires upon the later of 20 years from its effective date or the expiry of the last
patent licensed thereunder, unless otherwise terminated.
Unless otherwise terminated, the Isis agreements for OGX-011 and OGX-427 will continue for each
product until the later of 10 years after the date of the first commercial product sale, or the
expiration of the last to expire of any patents required to be licensed in order to use or sell the
product, unless OncoGenex Technologies abandons either OGX-011 or OGX-427 and Isis does not elect
to unilaterally continue development. The Isis agreement for OGX-225 will continue into perpetuity
unless OncoGenex Technologies abandons the product and Isis does not elect to unilaterally continue
development.
Under the Second Isis Amendment, the Company is the subject of a change of control with a third
party, where the surviving company immediately following such change of control has the right to
develop and sell the product, then (i) a milestone payment of $20 million will be due and payable
to Isis 21 days following the first commercial sale of the product in the United States; and (ii)
unless such surviving entity had previously sublicensed the product and a royalty rate payable to
Isis by the Company has been established, the applicable royalty rate payable to Isis will
thereafter be the maximum amount payable under the Companys agreement with Isis. Any non-royalty
milestone amounts previously paid will be credited toward the $20 million milestone if not already
paid. As a result of the $10 million milestone payment payable to Isis in relation to the
Collaboration Agreement, the remaining amount owing in the event of change of control discussed
above is a maximum of $10 million. As the Company has now licensed the product to Teva and
established a royalty rate payable to Isis, no royalty rate adjustments would apply if Teva
acquires the Company and is the surviving company.
Bayer HealthCare LLC
On August 7, 2008, Sonus completed an exclusive in-licensing agreement with Bayer for the right to
develop, commercialize or sublicense a family of compounds known as caspase activators presently in
preclinical research. Under terms of the agreement, Sonus was granted exclusive rights to develop
two core compounds for all prophylactic and therapeutic uses in humans. Additionally, Sonus was
granted rights to all other non-core compounds covered under the patents for use in oncology.
Under the terms of the agreement, Bayer received an upfront license fee of $450,000. OncoGenex will
make Anniversary Payments, with an initial payment of $100,000. The payments will increase by
$25,000 each year until the initiation of the first phase 3 clinical trial, at which point the
Anniversary Payments reset to $100,000 each year and increase by $25,000 until the Company achieves
either the first New Drug Application filing in the United States or the European Union. OncoGenex
is obligated to pay royalties on net future product sales in
addition to aggregate milestone payments of up to $14,000,000 for clinical development and regulatory milestones. No milestone
payments are triggered prior to the initiation of a phase 3 clinical trial. OncoGenex has the
option to terminate this contract upon 60 days written notice to Bayer.
60
Lease Arrangements
The Company has an operating lease agreement for office space in Vancouver, Canada, which expires
in March 2011.
Future minimum annual lease payments under the Vancouver lease are as follows (in thousands):
| |
|
|
|
|
2010 |
|
$ |
181 |
|
2011 |
|
|
45 |
|
|
|
|
|
Total |
|
$ |
226 |
|
In November 2006, prior to the Arrangement, Sonus entered into a non-cancellable operating lease
agreement for office space in Bothell, Washington, expiring in 2017. In connection with the new
lease, Sonus was required to provide a cash security deposit of approximately $497,000, which is
included in Other Long Term Assets. In addition, the lease stipulates the Company must issue a
standby letter of credit for approximately $500,000 which is expected to be issued during 2010. The
Company is currently in the process of evaluating opportunities to exit or sublet portions of the
leased space and has recorded a liability in the excess facilities lease charge of $4,645,000 as at
December 31, 2009.
If the Company is unable to exit or sublet portions of this leased space, the future minimum annual
lease payments are as follows (in thousands):
| |
|
|
|
|
2010 |
|
$ |
1,995 |
|
2011 |
|
|
2,055 |
|
2012 |
|
|
2,117 |
|
2013 |
|
|
2,180 |
|
2014 |
|
|
2,245 |
|
remainder |
|
|
7,150 |
|
|
|
|
|
Total |
|
$ |
17,742 |
|
Consolidated rent expense relating to both the Vancouver, Canada and Bothell, Washington offices
for years ended December 31, 2009, 2008, and 2007 was $2,408,000, $801,000, and $220,000
respectively.
Guarantees and Indemnifications
OncoGenex indemnifies its officers and directors for certain events or occurrences, subject to
certain limits, while the officer or director is or was serving at our request in such capacity.
The term of the indemnification period is equal to the officers or directors lifetime.
The maximum amount of potential future indemnification is unlimited; however, we have obtained
director and officer insurance that limits our exposure and may enable it to recover a portion of
any future amounts paid. We believe that the fair value of these indemnification obligations is
minimal. Accordingly, we have not recognized any liabilities relating to these obligations as of
December 31, 2009.
We have certain agreements with certain organizations with which we do business that contain
indemnification provisions pursuant to which we typically agree to indemnify the party against
certain types of third-party claims. We accrue for known indemnification issues when a loss is
probable and can be reasonably estimated. There were no accruals for or expenses related to
indemnification issues for any period presented.
61
Material Changes in Financial Condition
| |
|
|
|
|
|
|
|
|
| (in thousands) |
|
December 31,
2009 |
|
|
December 31,
2008 |
|
Total Assets |
|
$ |
68,980 |
|
|
$ |
14,790 |
|
Total Liabilities |
|
$ |
46,021 |
|
|
$ |
4,083 |
|
Total Equity |
|
$ |
22,959 |
|
|
$ |
10,707 |
|
The increase in assets from December 31, 2008 primarily relates to increase in cash, cash
equivalents and marketable securities following the Collaboration Agreement with Teva. The
increase in liabilities from December 31, 2008 relates to deferred collaboration revenue and
milestone payments owing to Isis and UBC resulting from our Collaboration Agreement with Teva. The
increase in equity relates predominantly to the issuance of shares through private placements of
our common stock and shares sold to Teva.
Critical Accounting Policies and Estimates
Significant Accounting Policies
Revenue Recognition
Revenue recognized to date is attributable solely to the upfront payment the Company received in
the fourth quarter of 2009 pursuant to its Collaboration Agreement with Teva. For a description of
the Collaboration Agreement, see note 4 in the Notes to Financial Statements for further details on
our collaboration with Teva.
The Collaboration Agreement contains multiple elements and deliverables, and requires evaluation
pursuant to ASC 605-25, Multiple-Element Arrangements (ASC 605-25). The Company evaluated the
facts and circumstances of the Collaboration Agreement to determine whether it had obligations
constituting deliverables under ASC 605-25. The Company concluded that it had multiple deliverables
under the Collaboration Agreement, including deliverables relating to the grant of a technology
license, and performance of manufacturing, regulatory and clinical development services in the U.S.
and Canada, and estimated that the period in which the Company would perform those deliverables
began in the fourth quarter of 2009 and will be completed in 2013. Because the Company was able to
establish vendor specific objective evidence (VSOE) of the fair value of the maintenance,
regulatory, and clinical services, the Company concluded that these deliverables should be
accounted as separate units of accounting under ASC 605-25. In establishing VSOE for the
manufacturing, regulatory, and clinical development services, management relied on rates charged by
other service providers providing similar development services.
The Company will recognize $30 million allocated to the manufacturing, regulatory and clinical
development services element as revenue over the estimated performance period, using the
proportional performance model. Estimation of the performance period of the Companys deliverables
required the use of managements judgement. Significant factors considered in managements
evaluation of the estimated performance period include, but are not limited to its experience,
along with Tevas experience, in conducting clinical development
activities. The Company will
review the estimated duration of its performance period on a quarterly basis and make any
appropriate adjustments on a prospective basis. Future changes in estimates of the performance
period may materially impact the timing of the future revenue recognized under the Collaboration
Agreement.
Because management is not able to reliably estimate the fair value of the technology license, it
used the residual value approach to determine the amount of revenue to recognize. Based on this
approach, the Company recognized $22 million in 2009 relating to this element.
62
Under the Collaboration Agreement, the Company is entitled to receive up to $370 million upon the
achievement of developmental and commercial milestones. Management evaluated the nature of the
events triggering these contingent payments and concluded that these events constituted substantive
milestones. This conclusion was based primarily on the facts that each triggering event represents
a specific outcome that can be achieved only through successful performance by the Company of one
or more of its deliverables, and that achievement of each triggering event was subject to inherent
risk and uncertainty and would result in additional payments becoming due to the Company.
Management concluded that each of these milestones was substantive, based primarily on the facts
that the payments they trigger are non-refundable, that achievement of the milestone entails risk
and was not reasonably assured at inception of the Collaboration Agreement, that substantial effort
is required to complete each milestone, that the amount of each milestone payment is reasonable in
relation to the value created in achieving the milestone, that a substantial amount of time is
expected to pass between the upfront payment and the potential milestone payments, and that the
milestone payments, once received, relate solely to past performance. Based on the foregoing, the
Company will recognize any revenue from these milestone payments under the substantive milestone
method in the period in which the underlying triggering event occurs.
Under the Collaboration Agreement, the Company is also entitled to receive royalties on sales of
OGX-011 ranging from the mid-teens to the mid-twenties. The Company will recognize any revenue from
these events based on the revenue recognition criteria set forth in ASC 605, Revenue Recognition.
Based on those criteria, the Company considers these potential payments to be contingent revenues,
and will recognize them as revenue in the period in which the applicable contingency is resolved.
Under the Collaboration Agreement, the Company and Teva share certain OGX-011-related development
costs. The Company will be required to spend $30 million in direct and indirect development costs
such as full-time equivalent (FTE) reimbursement for time incurred by OncoGenex personnel for the
benefit of the OGX-011 development plan, such contribution to be funded by the upfront payment
provided by Teva as an advanced reimbursement for the Company Development Expenses. Teva will fund
all other expenses under the Clinical Development Plan (Clinical Development Plan). Once the
Company has fulfilled its requirement to spend $30 million in direct and indirect development
costs, including FTEs, Teva will, on a quarterly basis, reimburse all development expenses incurred
in accordance with the Clinical Development Plan. The Companys policy is to account for these
reimbursements as Collaboration Revenue.
Income Taxes
Income taxes are accounted for under the liability method. Deferred tax assets and liabilities are
recognized for the differences between the carrying values of assets and liabilities and their
respective income tax bases and for operating losses and tax credit carry forwards. A valuation
allowance is provided for the portion of deferred tax assets that is more likely than not to be
unrealized. Deferred tax assets and liabilities are measured using the enacted tax rates and laws.
Research and Development Costs
Research and development costs are expensed as incurred, net of related refundable investment tax
credits, with the exception of non-refundable advanced payments for goods or services to be used in
future research and development, which are capitalized in accordance with ASC 730, Research and
Development and included within Other Assets.
Clinical trial expenses are a component of research and development costs. These expenses include
fees paid to contract research organizations and investigators and other service providers, which
conduct certain product development activities on our behalf. The Company uses an accrual basis of
accounting, based upon estimates of the amount of service completed. In the event payments differ
from the amount of service completed, prepaid expense or accrued liabilities amounts are adjusted on the balance sheet. These
expenses are based on estimates of the work performed under service agreements, milestones
achieved, patient Enrollment and experience with similar contracts. The Company monitors each of
these factors to the extent possible and adjusts estimates accordingly.
63
Stock-Based Compensation
Effective January 1, 2006, the Company adopted the fair value recognition provisions of the ASC
718, Stock Compensation, using the modified prospective method with respect to options granted to
employees and directors. Under this transition method, compensation cost is recognized in the
financial statements beginning with the effective date for all share-based payments granted after
January 1, 2006 and for all awards granted prior to but not yet vested as of January 1, 2006. The
expense is amortized on a straight-line basis over the graded vesting period. Short-Term
Investments
Short-term investments consist of financial instruments purchased with an original maturity of
greater than three months and less than one year. The Company considers its short-term investments
as available-for-sale and they are carried at market value with unrealized gains and losses, if
any, reported as accumulated other comprehensive income or loss, which is a separate component of
shareholders equity. Realized gains and losses on the sale of these securities are recognized in
net loss. The cost of investments sold is based on the specific identification method.
Fair value of financial instruments
The fair value of the Companys cash equivalents and marketable securities is based on quoted
market prices and trade data for comparable securities. Other financial instruments including
amounts receivable, accounts payable and accrued liabilities, are carried at cost, which the
Company believes approximates fair value because of the short-term maturities of these instruments.
Recent accounting pronouncements
There were no recent accounting pronouncements which the Company expects would have any impact on
the consolidated financial position, results of operations or cash flows.
ITEM 7A. QUANTITATIVE AND QUALITATIVE DISCLOSURES ABOUT MARKET RISK
Interest Rate Risk
We invest our cash in a variety of financial instruments, primarily in short-term bank deposits,
money market funds, and domestic and foreign commercial paper and government securities. These
investments are denominated in U.S. dollars and are subject to interest rate risk, and could
decline in value if interest rates fluctuate. Our investment portfolio includes only marketable
securities with active secondary or resale markets to help ensure portfolio liquidity. Due to the
conservative nature of these instruments, we do not believe that we have a material exposure to
interest rate risk. For example, if market rates hypothetically increase immediately and uniformly
by 100 basis points from levels at December 31, 2009, the decline in the fair value of our
investment portfolio would not be material.
Foreign Currency Exchange Risk
We are exposed to risks associated with foreign currency transactions on certain contracts and
payroll expenses related to our Canadian subsidiary, OncoGenex Technologies, denominated in
Canadian dollars and we have not hedged these amounts. As our unhedged foreign currency
transactions fluctuate, our earnings might be negatively affected. Accordingly, changes in the
value of the U.S. dollar relative to the Canadian dollar might have an adverse effect on our
reported results of operations and financial condition, and fluctuations in exchange rates might
harm our reported results and accounts from period to period.
64
ITEM 8. FINANCIAL STATEMENTS AND SUPPLEMENTARY DATA
INDEX TO FINANCIAL STATEMENTS:
65
Report of Independent Registered Public Accounting Firm
To the Board of Directors and Shareholders of
OncoGenex Pharmaceuticals, Inc.
We have audited the accompanying consolidated balance sheets of OncoGenex Pharmaceuticals, Inc.
(the Company) as of December 31, 2009 and 2008, and the related consolidated statements of loss,
shareholders equity, and cash flows for each of the three years in the period ended December 31,
2009. These financial statements are the responsibility of the Companys management. Our
responsibility is to express an opinion on these financial statements based on our audits.
We conducted our audits in accordance with the standards of the Public Company Accounting Oversight
Board (United States). Those standards require that we plan and perform the audit to obtain
reasonable assurance about whether the financial statements are free of material misstatement. An
audit includes examining, on a test basis, evidence supporting the amounts and disclosures in the
financial statements. An audit also includes assessing the accounting principles used and
significant estimates made by management, as well as evaluating the overall financial statement
presentation. We believe that our audits provide a reasonable basis for our opinion.
In our opinion, the financial statements referred to above present fairly, in all material
respects, the consolidated financial position of OncoGenex Pharmaceuticals, Inc. at December 31,
2009 and 2008, and the consolidated results of its operations and its cash flows for each of the
three years in the period ended December 31, 2009, in conformity with U.S. generally accepted
accounting principles.
We also have audited, in accordance with the standards of the Public Company Accounting Oversight
Board (United States), OncoGenex Pharmaceuticals, Inc.s internal control over financial reporting
as of December 31, 2009, based on criteria established in Internal Control-Integrated Framework
issued by the Committee of Sponsoring Organizations of the Treadway Commission and our report dated
March 8, 2010 expressed an unqualified opinion thereon.
| |
|
|
Vancouver, Canada,
March 8, 2010
|
|
/s/ ERNST & YOUNG LLP |
66
Report of Independent Registered Public Accounting Firm
To the Board of Directors and Shareholders of
OncoGenex Pharmaceuticals, Inc.
We have audited OncoGenex Pharmaceuticals, Inc.s (the Company) internal control over financial
reporting as of December 31, 2009, based on criteria established in Internal ControlIntegrated
Framework issued by the Committee of Sponsoring Organizations of the Treadway Commission (the
COSO criteria). The Companys management is responsible for maintaining effective internal
control over financial reporting, and for its assessment of the effectiveness of internal control
over financial reporting included in the accompanying Managements Report on Internal Control over
Financial Reporting. Our responsibility is to express an opinion on the Companys internal control
over financial reporting based on our audit.
We conducted our audit in accordance with the standards of the Public Company Accounting Oversight
Board (United States). Those standards require that we plan and perform the audit to obtain
reasonable assurance about whether effective internal control over financial reporting was
maintained in all material respects. Our audit included obtaining an understanding of internal
control over financial reporting, assessing the risk that a material weakness exists, testing and
evaluating the design and operating effectiveness of internal control based on the assessed risk,
and performing such other procedures as we considered necessary in the circumstances. We believe
that our audit provides a reasonable basis for our opinion.
A companys internal control over financial reporting is a process designed to provide reasonable
assurance regarding the reliability of financial reporting and the preparation of financial
statements for external purposes in accordance with generally accepted accounting principles. A
companys internal control over financial reporting includes those policies and procedures that (1)
pertain to the maintenance of records that, in reasonable detail, accurately and fairly reflect the
transactions and dispositions of the assets of the company; (2) provide reasonable assurance that
transactions are recorded as necessary to permit preparation of financial statements in accordance
with generally accepted accounting principles, and that receipts and expenditures of the company
are being made only in accordance with authorizations of management and directors of the company;
and (3) provide reasonable assurance regarding prevention or timely detection of unauthorized
acquisition, use, or disposition of the companys assets that could have a material effect on the
financial statements.
Because of its inherent limitations, internal control over financial reporting may not prevent or
detect misstatements. Also, projections of any evaluation of effectiveness to future periods are
subject to the risk that controls may become inadequate because of changes in conditions, or that
the degree of compliance with the policies or procedures may deteriorate.
In our opinion, the Company maintained, in all material respects, effective internal control over
financial reporting as of December 31, 2009, based on the COSO criteria.
We also have audited, in accordance with the standards of the Public Company Accounting Oversight
Board (United States), the consolidated balance sheets of the Company as of December 31, 2009 and
2008, and the related consolidated statements of loss, shareholders equity, and cash flows for
each of the three years in the period ended December 31, 2009 and our report dated March 8, 2010
expressed an unqualified opinion thereon.
|
|
Vancouver, Canada
March 8, 2010 |
|
/s/ ERNST & YOUNG LLP |
67
OncoGenex Pharmaceuticals, Inc.
Consolidated Balance Sheets
(In thousands of U.S. dollars)
| |
|
|
|
|
|
|
|
|
| |
|
December 31, |
|
|
December 31, |
|
| |
|
2009 |
|
|
2008 |
|
| |
|
$ |
|
|
$ |
|
|
|
|
|
|
|
|
|
|
ASSETS |
|
|
|
|
|
|
|
|
Current |
|
|
|
|
|
|
|
|
Cash and cash equivalents |
|
|
62,051 |
|
|
|
7,618 |
|
Short-term investments [note 6]
|
|
|
2,517 |
|
|
|
4,801 |
|
Amounts receivable [note 4]
|
|
|
3,109 |
|
|
|
153 |
|
Investment tax credit recoverable |
|
|
|
|
|
|
1,090 |
|
Prepaid expenses |
|
|
722 |
|
|
|
587 |
|
|
|
|
|
|
|
|
Total current assets |
|
|
68,399 |
|
|
|
14,249 |
|
Property and equipment, net [note 7]
|
|
|
72 |
|
|
|
44 |
|
Other assets |
|
|
509 |
|
|
|
497 |
|
|
|
|
|
|
|
|
Total assets |
|
|
68,980 |
|
|
|
14,790 |
|
|
|
|
|
|
|
|
|
|
LIABILITIES AND SHAREHOLDERS EQUITY |
|
|
|
|
|
|
|
|
Current |
|
|
|
|
|
|
|
|
Accounts payable and accrued liabilities |
|
|
14,453 |
|
|
|
2,252 |
|
Deferred Collaboration Revenue |
|
|
10,000 |
|
|
|
|
|
Current portion of long-term obligations [note 8]
|
|
|
1,328 |
|
|
|
632 |
|
|
|
|
|
|
|
|
Total current liabilities |
|
|
25,781 |
|
|
|
2,884 |
|
Deferred Collaboration Revenue, net of current |
|
|
16,528 |
|
|
|
|
|
Long-term obligation, less current portion [note 8]
|
|
|
3,712 |
|
|
|
1,199 |
|
|
|
|
|
|
|
|
Total liabilities |
|
|
46,021 |
|
|
|
4,083 |
|
Commitments and contingencies [note 13]
|
|
|
|
|
|
|
|
|
Shareholders equity: |
|
|
|
|
|
|
|
|
Common shares: |
|
|
|
|
|
|
|
|
$0.001 par value 11,019,930 shares authorized and
6,324,033 issued and
outstanding at December 31, 2009 |
|
|
6 |
|
|
|
6 |
|
Additional paid-in capital |
|
|
73,798 |
|
|
|
56,070 |
|
Accumulated deficit |
|
|
(53,485 |
) |
|
|
(48,009 |
) |
Accumulated other comprehensive income |
|
|
2,640 |
|
|
|
2,640 |
|
|
|
|
|
|
|
|
Total shareholders equity |
|
|
22,959 |
|
|
|
10,707 |
|
|
|
|
|
|
|
|
Total liabilities and shareholders equity |
|
|
68,980 |
|
|
|
14,790 |
|
Subsequent events [note 16] |
|
|
|
|
|
|
|
|
68
OncoGenex Pharmaceuticals, Inc.
Consolidated Statements of Loss
(In thousands of U.S. dollars, except share and per share amounts)
| |
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
Years Ended |
|
| |
|
December 31, |
|
| |
|
2009 |
|
|
2008 |
|
|
2007 |
|
| |
|
$ |
|
|
$ |
|
|
$ |
|
COLLABORATION REVENUE |
|
|
25,539 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
EXPENSES |
|
|
|
|
|
|
|
|
|
|
|
|
Research and development |
|
|
24,160 |
|
|
|
7,819 |
|
|
|
4,135 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
General and administrative |
|
|
3,961 |
|
|
|
3,293 |
|
|
|
3,540 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total expenses |
|
|
28,121 |
|
|
|
11,112 |
|
|
|
7,675 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
OTHER INCOME (EXPENSE) |
|
|
|
|
|
|
|
|
|
|
|
|
Interest income |
|
|
47 |
|
|
|
210 |
|
|
|
177 |
|
Other |
|
|
70 |
|
|
|
211 |
|
|
|
(325 |
) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total other income (expense) |
|
|
117 |
|
|
|
421 |
|
|
|
(148 |
) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Loss for the period before taxes and
extraordinary gain |
|
|
(2,465 |
) |
|
|
(10,691 |
) |
|
|
(7,823 |
) |
Income tax expense (recovery) [note 10]
|
|
|
3,011 |
|
|
|
(2,059 |
) |
|
|
713 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Loss before extraordinary gain |
|
|
(5,476 |
) |
|
|
(8,632 |
) |
|
|
(8,536 |
) |
Extraordinary gain [note 5]
|
|
|
|
|
|
|
4,428 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net loss |
|
|
(5,476 |
) |
|
|
(4,204 |
) |
|
|
(8,536 |
) |
Redeemable convertible preferred share accretion |
|
|
|
|
|
|
1,973 |
|
|
|
2,944 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Loss attributable to common shareholders |
|
|
(5,476 |
) |
|
|
(6,177 |
) |
|
|
(11,480 |
) |
|
|
|
|
|
|
|
|
|
|
|
|
|
Basic and diluted loss per common share
[note 11[h]]
|
|
|
(.95 |
) |
|
|
(3.38 |
) |
|
|
(96.63 |
) |
|
|
|
|
|
|
|
|
|
|
|
|
|
Weighted average number of common shares [note
11[h]]
|
|
|
5,766,850 |
|
|
|
1,829,276 |
|
|
|
118,801 |
|
|
|
|
|
|
|
|
|
|
|
See accompanying notes.
69
OncoGenex Pharmaceuticals, Inc.
Consolidated Statements of Shareholders Equity
(In thousands of U.S. dollars, except share amounts)
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
Accumulated |
|
|
|
|
|
|
|
|
|
|
Total |
|
| |
|
|
|
|
|
|
|
|
|
Other |
|
|
|
|
|
|
|
|
|
|
Shareholders |
|
| |
|
Common |
|
|
|
|
|
|
Comprehensive |
|
|
Comprehensive |
|
|
Accumulated |
|
|
Equity |
|
| |
|
Shares |
|
|
Amount |
|
|
Income (Loss) |
|
|
Income (Loss) |
|
|
deficit |
|
|
(Deficiency) |
|
| |
|
(in thousands of U.S. dollars, except share amounts) |
|
Balance, December 31, 2006 |
|
|
118,801 |
|
|
|
703 |
|
|
|
2,083 |
|
|
|
|
|
|
|
(30,352 |
) |
|
|
(27,566 |
) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Stock-based compensation expense |
|
|
|
|
|
|
263 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
263 |
|
Cumulative translation adjustment from application of US
dollar reporting |
|
|
|
|
|
|
|
|
|
|
557 |
|
|
|
557 |
|
|
|
|
|
|
|
557 |
|
Reclassification of unrealized loss on marketable securities |
|
|
|
|
|
|
|
|
|
|
2 |
|
|
|
2 |
|
|
|
|
|
|
|
2 |
|
Unrealized gain on marketable securities |
|
|
|
|
|
|
|
|
|
|
1 |
|
|
|
1 |
|
|
|
|
|
|
|
1 |
|
Redeemable convertible preferred share accretion |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(2,944 |
) |
|
|
(2,944 |
) |
Loss for the year |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(8,536 |
) |
|
|
(8,536 |
) |
|
|
(8,536 |
) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Comprehensive loss for the year |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(7,976 |
) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Balance, December 31, 2007 |
|
|
118,801 |
|
|
|
966 |
|
|
|
2,643 |
|
|
|
|
|
|
|
(41,832 |
) |
|
|
(38,223 |
) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Stock-based compensation expense |
|
|
|
|
|
|
174 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
174 |
|
Shares held by Sonus shareholders |
|
|
2,059,898 |
|
|
|
10,456 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
10,456 |
|
Shares issued in exchange for convertible debentures |
|
|
1,036,586 |
|
|
|
5,012 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
5,012 |
|
Shares issued in exchange for preferred shares |
|
|
905,131 |
|
|
|
39,345 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
39,345 |
|
Escrow shares released on achievement of milestones |
|
|
1,388,875 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Stock option exercises |
|
|
34,601 |
|
|
|
122 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
122 |
|
Issuance of common stock under employee benefit plans |
|
|
222 |
|
|
|
1 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
1 |
|
Cumulative translation adjustment from application of US
dollar reporting |
|
|
|
|
|
|
|
|
|
|
(3 |
) |
|
|
(3 |
) |
|
|
|
|
|
|
(3 |
) |
Redeemable convertible preferred share accretion |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(1,973 |
) |
|
|
(1,973 |
) |
Loss for the period |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(4,204 |
) |
|
|
(4,204 |
) |
|
|
(4,204 |
) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Comprehensive loss for the period |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(4,207 |
) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Balance, December 31, 2008 |
|
|
5,544,114 |
|
|
|
56,076 |
|
|
|
2,640 |
|
|
|
|
|
|
|
(48,009 |
) |
|
|
10,707 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Stock-based compensation expense |
|
|
|
|
|
|
380 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
380 |
|
Shares issued in July 2009 financing |
|
|
475,000 |
|
|
|
9,304 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
9,304 |
|
Shares issued in Teva share purchase agreement |
|
|
267,531 |
|
|
|
7,903 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
7,903 |
|
Stock option exercises |
|
|
37,388 |
|
|
|
141 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
141 |
|
Loss for the period |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(5,476 |
) |
|
|
(5,476 |
) |
|
|
(5,476 |
) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Comprehensive loss for the period |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(5,476 |
) |
|
|
|
|
|
|
|
|
Balance, December 31, 2009 |
|
|
6,324,033 |
|
|
|
73,804 |
|
|
|
2,640 |
|
|
|
|
|
|
|
(53,485 |
) |
|
|
22,959 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
70
OncoGenex Pharmaceuticals, Inc.
Consolidated Statements of Cash Flows
(In thousands of U.S. dollars)
| |
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
Years ended |
|
| |
|
December 31, |
|
| |
|
2009 |
|
|
2008 |
|
|
2007 |
|
| |
|
$ |
|
|
$ |
|
|
$ |
|
OPERATING ACTIVITIES |
|
|
|
|
|
|
|
|
|
|
|
|
Loss for the period |
|
|
5,476 |
|
|
|
4,204 |
|
|
|
8,536 |
|
|
|
|
|
|
|
|
|
|
|
Add items not involving cash |
|
|
|
|
|
|
|
|
|
|
|
|
Extraordinary gain |
|
|
|
|
|
|
(4,428 |
) |
|
|
|
|
Depreciation and amortization |
|
|
50 |
|
|
|
89 |
|
|
|
83 |
|
Stock-based compensation [note 11[c]]
|
|
|
380 |
|
|
|
174 |
|
|
|
263 |
|
Accrued interest on convertible debenture [note 11]
|
|
|
|
|
|
|
313 |
|
|
|
193 |
|
Changes in non-cash working capital items |
|
|
|
|
|
|
|
|
|
|
|
|
Amounts receivable |
|
|
(2,955 |
) |
|
|
204 |
|
|
|
181 |
|
Investment tax credit recoverable |
|
|
1,090 |
|
|
|
646 |
|
|
|
(993 |
) |
Prepaid expenses |
|
|
(136 |
) |
|
|
|
|
|
|
(68 |
) |
Other assets |
|
|
(12 |
) |
|
|
(117 |
) |
|
|
(1 |
) |
Accounts payable and accrued liabilities |
|
|
12,200 |
|
|
|
(2,244 |
) |
|
|
(2 |
) |
Lease obligation |
|
|
3,210 |
|
|
|
(253 |
) |
|
|
|
|
Deferred collaboration revenue |
|
|
26,527 |
|
|
|
|
|
|
|
|
|
Taxes payable on preferred shares |
|
|
|
|
|
|
(2,487 |
) |
|
|
1,001 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash provided by (used in) operating activities |
|
|
34,878 |
|
|
|
(12,307 |
) |
|
|
(7,879 |
) |
|
|
|
|
|
|
|
|
|
|
|
|
|
FINANCING ACTIVITIES |
|
|
|
|
|
|
|
|
|
|
|
|
Cash paid on fractional shares eliminated on reverse share split |
|
|
|
|
|
|
(3 |
) |
|
|
|
|
Proceeds from issuance of common stock under stock option and
employee purchase plans |
|
|
141 |
|
|
|
124 |
|
|
|
|
|
Issuance of common shares, net of share issue costs |
|
|
17,206 |
|
|
|
|
|
|
|
|
|
Issuance of convertible debentures net of issue costs |
|
|
|
|
|
|
|
|
|
|
4,442 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash provided by financing activities |
|
|
17,347 |
|
|
|
121 |
|
|
|
4,442 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
INVESTING ACTIVITIES |
|
|
|
|
|
|
|
|
|
|
|
|
Purchase of investments |
|
|
(4,036 |
) |
|
|
(4,843 |
) |
|
|
(6,763 |
) |
Proceeds from sale of investments |
|
|
6,280 |
|
|
|
15,276 |
|
|
|
13,058 |
|
Purchase of property and equipment |
|
|
(15 |
) |
|
|
(3 |
) |
|
|
(17 |
) |
Cash received on reverse takeover of Sonus |
|
|
|
|
|
|
5,464 |
|
|
|
|
|
Transaction fees on reverse takeover of Sonus |
|
|
|
|
|
|
(807 |
) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash provided by investing activities |
|
|
2,229 |
|
|
|
15,087 |
|
|
|
6,278 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Effect of exchange rate changes on cash |
|
|
(21 |
) |
|
|
90 |
|
|
|
(68 |
) |
Increase in cash and cash equivalents during the period |
|
|
54,433 |
|
|
|
2,992 |
|
|
|
2,773 |
|
Cash and cash equivalents, beginning of the period |
|
|
7,618 |
|
|
|
4,626 |
|
|
|
1,853 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash and cash equivalents, end of the period |
|
|
62,051 |
|
|
|
7,618 |
|
|
|
4,626 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Supplemental cash flow information |
|
|
|
|
|
|
|
|
|
|
|
|
Property and equipment acquired under lease obligation |
|
|
65 |
|
|
|
|
|
|
|
|
|
See accompanying notes.
71
OncoGenex Pharmaceuticals, Inc.
Notes to Consolidated Financial Statements
1. NATURE OF BUSINESS AND BASIS OF PRESENTATION
OncoGenex Pharmaceuticals, Inc. (the Company or OncoGenex) is committed to the development and
commercialization of new therapies that address treatment resistance in cancer patients. The
Company was incorporated in the state of Delaware and, together with its subsidiaries, has a
facility in Bothell, Washington for administrative, clinical and regulatory operations and an
office in Vancouver, British Columbia (Canada) for administrative, pre-clinical and
manufacturing-related operations.
On August 21, 2008, Sonus Pharmaceuticals, Inc. (Sonus) completed a transaction (the
Arrangement) with OncoGenex Technologies Inc., (OncoGenex Technologies) whereby Sonus acquired
all of the outstanding preferred shares, common shares and convertible debentures of OncoGenex
Technologies. Sonus changed its name to OncoGenex Pharmaceuticals, Inc. and was listed on the
Nasdaq Capital Market under the ticker symbol OGXI. These consolidated financial statements account
for the Arrangement between Sonus and OncoGenex Technologies as a reverse acquisition, whereby
OncoGenex Technologies is deemed to be the acquiring entity from an accounting perspective.
During the year ended December 31, 2009, the Company exited the development stage. Previously from
its inception, the Company was a development stage company in accordance with Accounting Standards
Codification (ASC) 915, Accounting and Reporting by Development Stage Enterprises.
Basis of Presentation
The accompanying consolidated financial statements have been prepared in accordance with accounting
principles generally accepted in the United States. In the opinion of management, all adjustments
(consisting of normal recurring adjustments) considered necessary for a fair presentation of the
financial position and results of operations of OncoGenex have been included. The consolidated
financial statements include the accounts of OncoGenex Pharmaceuticals, Inc. and our wholly owned
subsidiaries, OncoGenex Technologies and OncoGenex, Inc. OncoGenex, Inc. ceased operations in 2009
and was subsequently dissolved. All intercompany balances and transactions have been eliminated.
Liquidity
The Company has historically experienced recurring losses from operations that have generated an
accumulated deficit of $53.5 million through December 31, 2009. At December 31, 2009, the Company
had cash, cash equivalents and short-term investments of $64.6 million, and working capital of
$42.6 million.
Based on the current forecasted cash needs for the Company, management believes that existing cash,
cash equivalents and short-term investments will be sufficient to fund expected operations into
2012.
2. ACCOUNTING POLICIES
Significant Accounting Policies
Use of Estimates
The preparation of consolidated financial statements in conformity with United States generally
accepted accounting principles requires management to make estimates and assumptions that affect
the amounts reported in the consolidated financial statements and notes thereto. Actual results
could differ from those estimates. Estimates and assumptions principally relate to the performance
period of the Companys deliverables under its Collaboration Agreement with Teva Pharmaceutical
Industries Ltd. (Teva), estimates of the fair value and forfeiture rates of stock options issued
to employees and consultants, the resolution of uncertain tax positions and estimates of the fair
value of our excess lease facility liability.
72
Cash Equivalents
The Company considers all highly liquid investments with an original maturity of three months or
less to be cash equivalents, which the Company considers as available for sale and are carried at
market value with unrealized gains and losses, if any, reported as accumulated other comprehensive
income or loss, which is a separate component of shareholders equity.
Short-Term Investments
Short-term investments consist of financial instruments purchased with an original maturity of
greater than three months and less than one year. The Company considers its short-term investments
as available-for-sale and they are carried at market value with unrealized gains and losses, if
any, reported as accumulated other comprehensive income or loss, which is a separate component of
shareholders equity (deficiency). Realized gains and losses on the sale of these securities are
recognized in net income or loss. The cost of investments sold is based on the specific
identification method.
Fair value of financial instruments
The fair value of the Companys cash equivalents and marketable securities is based on quoted
market prices and trade data for comparable securities. Other financial instruments including amounts receivable, accounts payable and
accrued liabilities, are carried at cost, which the Company believes approximates fair value
because of the short-term maturities of these instruments.
Intellectual Property
The costs of acquiring intellectual property rights to be used in the research and development
process, including licensing fees and milestone payments, are charged to research and development
expense as incurred in situations where the Company has not identified an alternative future use
for the acquired rights, and are capitalized in situations where it has identified an alternative
future use. No costs associated with acquiring intellectual property rights have been capitalized
to date. Legal and other costs of prosecuting and maintaining intellectual property rights are
expensed as incurred.
Revenue Recognition
Revenue recognized to date is attributable solely to the upfront payment the Company received in
the fourth quarter of 2009 pursuant to its Collaboration Agreement with Teva. For a description of
the Collaboration Agreement, see Note 4.
The Collaboration Agreement contains multiple elements and deliverables, and requires evaluation
pursuant to ASC 605-25, Multiple-Element Arrangements (ASC 605-25). The Company evaluated the
facts and circumstances of the Collaboration Agreement to determine whether it had obligations
constituting deliverables under ASC 605-25. The Company concluded that it had multiple deliverables
under the Collaboration Agreement, including deliverables relating to the grant of a technology
license, and performance of manufacturing, regulatory and clinical development services in the U.S.
and Canada, and estimated that the period in which the Company would perform those deliverables
began in the fourth quarter of 2009 and will be completed in 2013. Because the Company was able to
establish vendor specific objective evidence (VSOE) of the fair value of the maintenance,
regulatory, and clinical services, the Company concluded that these deliverables should be
accounted as separate units of accounting under ASC 605-25. In establishing VSOE for the
manufacturing, regulatory, and clinical development services, management relied on rates charged by
other service providers providing similar development services.
73
The Company will recognize $30 million allocated to the manufacturing, regulatory and clinical
development services element as revenue over the estimated performance period, using the
proportional performance model. Estimation of the performance period of the Companys deliverables
required the use of managements judgement. Significant factors considered in managements
evaluation of the estimated performance period include, but are not limited to its experience,
along with Tevas experience, in conducting clinical development activities. The Company will
review the estimated duration of its performance period on a quarterly basis and make any
appropriate adjustments on a prospective basis. Future changes in estimates of the performance
period may materially impact the timing of the future revenue recognized under the Collaboration
Agreement.
Because management is not able to reliably estimate the fair value of the technology license, it
used the residual value approach to determine the amount of revenue to recognize. Based on this
approach, the Company recognized $22 million in 2009 relating to this element.
Under the Collaboration Agreement, the Company is entitled to receive up to $370 million upon the
achievement of developmental and commercial milestones. Management evaluated the nature of the
events triggering these contingent payments and concluded that these events constituted substantive
milestones. This conclusion was based primarily on the facts that each triggering event represents
a specific outcome that can be achieved only through successful performance by the Company of one
or more of its deliverables, and that achievement of each triggering event was subject to inherent
risk and uncertainty and would result in additional payments becoming due to the Company.
Management concluded that each of these milestones was substantive, based primarily on the facts
that the payments they trigger are non-refundable, that achievement of the milestone entails risk
and was not reasonably assured at inception of the Collaboration Agreement, that substantial effort
is required to complete each milestone, that the amount of each milestone payment is reasonable in
relation to the value created in achieving the milestone, that a substantial amount of time is
expected to pass between the upfront payment and the potential milestone payments, and that the
milestone payments, once received, relate solely to past performance. Based on the foregoing, the
Company will recognize any revenue from these milestone payments under the substantive milestone
method in the period in which the underlying triggering event occurs.
Under the Collaboration Agreement, the Company is also entitled to receive royalties on sales of
OGX-011 ranging from the mid-teens to the mid-twenties. The Company will recognize any revenue from
these events based on the revenue recognition criteria set forth in ASC 605, Revenue Recognition.
Based on those criteria, the Company considers these potential payments to be contingent revenues,
and will recognize them as revenue in the period in which the applicable contingency is resolved.
Under the Collaboration Agreement, the Company and Teva share certain OGX-011-related development
costs. The Company will be required to spend $30 million in direct and indirect development costs
such as full-time equivalent (FTE) reimbursement for time incurred by OncoGenex personnel for the
benefit of the OGX-011 development plan, such contribution to be funded by the upfront payment
provided by Teva as an advanced reimbursement for the Company Development Expenses. Teva will fund
all other expenses under the Clinical Development Plan (Clinical Development Plan). Once the
Company has fulfilled its requirement to spend $30 million in direct and indirect development
costs, including FTEs, Teva will, on a quarterly basis, reimburse all development expenses incurred
in accordance with the Clinical Development Plan. The Companys policy is to account for these
reimbursements as Collaboration Revenue.
Property and Equipment
Property and equipment assets are recorded at cost less accumulated amortization. Depreciation
expense on assets acquired under capital lease is recorded within depreciation expense.
Amortization is provided on a straight-line basis over the following periods:
| |
|
|
Computer equipment
|
|
3 years |
Computer software
|
|
3 years |
Furniture and fixtures
|
|
5 years |
Leasehold improvements
|
|
Over the term of the lease |
74
Reporting Currency and Foreign Currency Translation
Effective August 21, 2008, the Company changed its functional currency from the Canadian dollar to
the U.S. dollar. The Companys primary economic environment changed from Canada to the United
States following the acquisition of Sonus (note 5). This resulted in significant changes in
economic facts and circumstances that indicated that the functional currency had changed. The
Company accounted for the change in functional currency prospectively.
The consolidated financial statements of the Company for the year ended December 31, 2007 and for
the period of January 1, 2008 to August 20, 2008, which is based on the Canadian functional
currency, has been translated into the U.S. reporting currency using the current rate method as
required by ASC 830, Foreign Currency Matters, (ASC 830) as follows: assets and liabilities
using the rate of exchange prevailing at the balance sheet date; shareholders equity using the
applicable historic rate; and revenue and expenses using the monthly average rate of exchange.
Translation adjustments have been included as part of the accumulated other comprehensive income.
Income Taxes
Income taxes are accounted for under the liability method. Deferred tax assets and liabilities are
recognized for the differences between the carrying values of assets and liabilities and their
respective income tax bases and for operating losses and tax credit carry forwards. A valuation
allowance is provided for the portion of deferred tax assets that is more likely than not to be
unrealized. Deferred tax assets and liabilities are measured using the enacted tax rates and laws.
Scientific Research and Development Tax Credits
The benefits of tax credits for scientific research and development expenditures are recognized in
the year the qualifying expenditure is made provided there is reasonable assurance of
recoverability. The tax credits recorded are based on managements estimates of amounts expected to
be recovered and are subject to audit by taxation authorities. The refundable tax credit reduces
the carrying cost of expenditures for research and development expenses to which it relates. The
non-refundable tax credit reduces the tax provision. Following the completion of the Arrangement
(note 5) all qualifying expenditures are eligible for non-refundable tax credits only.
Research and Development Costs
Research and development costs are expensed as incurred, net of related refundable investment tax
credits, with the exception of non-refundable advanced payments for goods or services to be used in
future research and development, which are capitalized in accordance with ASC 730, Research and
Development and included within Other Assets.
Clinical trial expenses are a component of research and development costs. These expenses include
fees paid to contract research organizations and investigators and other service providers, which
conduct certain product development activities on our behalf. The Company uses an accrual basis of
accounting, based upon estimates of the amount of service completed. In the event payments differ
from the amount of service completed, prepaid expense or accrued liabilities amounts are adjusted
on the balance sheet. These expenses are based on estimates of the work performed under service
agreements, milestones achieved, patient Enrollment and experience with similar contracts. The
Company monitors each of these factors to the extent possible and adjusts estimates accordingly.
Stock-Based Compensation
Effective January 1, 2006, the Company adopted the fair value recognition provisions of the ASC
718, Stock Compensation, using the modified prospective method with respect to options granted to
employees and directors. Under this transition method, compensation cost is recognized in the
financial statements beginning with the effective date for all share-based payments granted after
January 1, 2006 and for all awards granted prior to but not yet vested as of January 1, 2006. The
expense is amortized on a straight-line basis over the graded vesting period.
75
Segment Information
The Company follows the requirements of ASC 280, Segment Reporting. The Company has one operating
segment, dedicated to the development and commercialization of new cancer therapies, with
operations located in Canada and the United States.
Comprehensive Income (Loss)
Comprehensive income (loss) is comprised of net income (loss) and other comprehensive income
(loss). Other comprehensive income (loss) consists of translation adjustments from the application
of U.S. dollar reporting until August 21, 2008 and unrealized gains and losses on the Companys
available-for-sale marketable securities. The Company has reported the components of comprehensive
loss in the statement of shareholders equity.
Loss per Common Share
Basic loss per common share is computed using the weighted average number of common shares
outstanding during the period adjusted to reflect the equivalent OncoGenex Pharmaceuticals shares
and equity structure. Prior to the completion of the Arrangement on August 21, 2008 the weighted
average number of common shares represents OncoGenex Technologies only. Diluted loss per common
share is computed in accordance with the treasury stock method which uses the weighted average
number of common shares outstanding during the period. The effect of potentially issuable common
shares from outstanding stock options and convertible preferred shares and debentures is
anti-dilutive for all periods presented.
Recently Adopted Accounting Policies
In November 2007, the FASB issued ASC 808, Collaborative Arrangements. ASC 808 requires
collaborators to present the results of activities for which they act as the principal on a gross
basis and report any payments received from (made to) other collaborators based on other applicable
GAAP or, in the absence of other applicable GAAP, based on analogy to authoritative accounting
literature or a reasonable, rational, and consistently applied accounting policy election.
Further, ASC 808 clarified that the determination of whether transactions within a collaborative
arrangement are part of a vendor-customer (or analogous) relationship. ASC 808 is effective for
fiscal years beginning after December 15, 2008. The Company implemented ASC 808 for the first time
for the Teva Collaboration Agreement.
In April 2009, the FASB issued amendments to ASC 320-10, Investment Debt and Equity Securities,
which modify the other-than-temporary impairment guidance for debt securities through increased
consistency in the timing of impairment recognition and enhanced disclosures related to the credit
and noncredit components of impaired debt securities that are not expected to be sold. In addition,
increased disclosures are required for both debt and equity securities regarding expected cash
flows, credit losses, and an aging of securities with unrealized losses. The amendments to ASC
320-10 become effective for interim and annual reporting periods that end after June 15, 2009, and
were adopted in our second quarter of 2009. The adoption of the amendments to ASC 320-10 has not
had a material impact on the consolidated financial position, results of operations or cash flows.
In April 2009, the FASB issued amendments to ASC 825-10, Financial Instruments, which require
fair value disclosures for financial instruments that are not reflected in the consolidated balance
sheets at fair value. Prior to the issuance these amendments to ASC 825-10, the fair values of
those assets and liabilities were disclosed only once each year. With the issuance the amendments
to ASC 825-10, are now required to disclose this information on a quarterly basis, providing
quantitative and qualitative information about fair value estimates for all financial instruments
not measured in the consolidated balance sheets at fair value. The amendments to ASC 825-10 become
effective for interim reporting periods that end after June 15, 2009, and were adopted in our
second quarter of 2009. The adoption of the amendments to ASC 825-10 has not had a material impact
on the consolidated financial position, results of operations or cash flows.
76
In April 2009, the FASB issued amendments to ASC 820-10, Fair Value Measurement and Disclosure,
which clarifies the methodology used to determine fair value when there is no active market or
where the price inputs being used represent distressed sales. The amendments to ASC 820-10 also
reaffirm the objective of fair value measurement, which is to reflect how much an asset would be
sold for in an orderly transaction. They also reaffirm the need to use judgment to determine if a
formerly active market has become inactive, as well as to determine fair values when markets have
become inactive. The amendments to ASC 820-10 which are applied prospectively, is effective for
interim and annual reporting periods ending after June 15, 2009, and was adopted in our second
quarter of 2009. The adoption of the amendments to ASC 820-10 has not had a material impact on the
consolidated financial position, results of operations or cash flows.
In May 2009, the FASB issued ASC 855, Subsequent Events. ASC 855 was issued in order to establish
principles and requirements for reviewing and reporting subsequent events and requires disclosure
of the date through which subsequent events are evaluated and whether the date corresponds with the
time at which the financial statements were available for issue (as defined) or were issued. ASC
855 is effective for interim reporting periods ending after June 15, 2009, and was adopted in our
second quarter of 2009. In accordance with ASC 855 it is the Companys policy to review and report
subsequent events up to the day prior to the issuance of the financial statements. The adoption of
ASC 855 has not had a material impact on the consolidated financial position, results of operations
or cash flows.
In June 2009, the FASB issued ASC 105, Generally Accepted Accounting Principles. ASC 105
establishes the FASB Accounting Standards Codification TM (Codification) as the source of
authoritative accounting principles recognized by the FASB to be applied by nongovernmental
entities in the preparation of financial statements in conformity with GAAP. Rules and interpretive
releases of the SEC under authority of federal securities laws are also sources of authoritative
GAAP for SEC registrants. The FASB will no longer issue new standards in the form of Statements,
FASB Staff Positions, or Emerging Issues
Task Force Abstracts; instead the FASB will issue Accounting Standards Updates. Accounting
Standards Updates will not be authoritative in their own right as they will only serve to update
the Codification. The issuance of ASC 105 and the Codification does not change GAAP. ASC 105 became
effective for the Company in the period ending December 31, 2009. The adoption of ASC 105 has not
had a material impact on our disclosure to the financial statements.
In October 2009, the FASB issued ASU 2009-13, which amends ASC Topic 605, Revenue Recognition.
Under this standard, management is no longer required to obtain vendor-specific objective evidence
or third party evidence of fair value for each deliverable in an arrangement with multiple
elements, and where evidence is not available we may now estimate the proportion of the selling
price attributable to each deliverable. The final consensus is effective for fiscal years beginning
on or after June 15, 2010. Entities can elect to apply Issue 08-1 prospectively to new or
materially modified arrangements after the effective date or retrospectively for all periods
presented. The Company does not anticipate that ASU 2009-13 will have any impact on the Companys
financial position or results of operations.
Recent Accounting Pronouncements
There were no recent accounting pronouncements which the Company expects would have any impact on
the consolidated financial position, results of operations or cash flows.
3. FINANCIAL INSTRUMENTS AND RISK
For certain of the companys financial instruments including cash, amounts receivable, and accounts
payable carrying values approximate fair value due to their short-term nature. The Companys cash
equivalents and short-term investments are recorded at fair value.
77
Financial risk is the risk to the Companys results of operations that arises from fluctuations in
interest rates and foreign exchange rates and the degree of volatility of these rates as well as
credit risk associated with the financial stability of the issuers of the financial instruments.
Foreign exchange rate risk arises as a portion of the Companys investments which finance
operations and a portion of the Companys expenses are denominated in other than U.S. dollars.
The Company invests its excess cash in accordance with investment guidelines, which limit the
credit exposure to any one financial institution or corporation other than securities issued by the
U.S. government. The guidelines also specify that the financial instruments are issued by
institutions with strong credit ratings. These securities generally mature within one year or less
and in some cases are not collateralized. At December 31, 2009 the average days to maturity of the
Companys portfolio of cash equivalents and marketable securities was 54 days. The Company does not
use derivative instruments to hedge against any of these financial risks.
4. COLLABORATION AGREEMENT
On December 20, 2009, the Company, through its wholly-owned subsidiary, OncoGenex Technologies
Inc., entered into a Collaboration Agreement with Teva for the development and global
commercialization of OGX-011 (and related compounds), a pharmaceutical compound designed to inhibit
the production of clusterin, a protein we believe associated with cancer treatment resistance.
Under the Collaboration Agreement, Teva paid the Company upfront payments in the aggregate amount
of $50 million, will pay up to $370 million upon the achievement of developmental and commercial
milestones and will pay royalties at percentage rates ranging from the mid-teens to mid-twenties on
net sales, depending on aggregate annual net sales of the Licensed Product. $3 million of the
upfront payments was paid to us subsequent to year end and is included in Amounts Receivable as of
December 31, 2009. The Company may be required to remit withholdings taxes to the Israeli Tax
Authority of up to $3 million. As a result, we have a recorded an Income Tax Expense of $3 million,
which is also included within Accounts Payable and Accrued Liabilities as at December 31, 2009.
On the same date, the Company and Teva also entered into a stock purchase agreement (the Stock
Purchase Agreement) pursuant to which Teva made an additional $10 million equity investment in the
Company at a 20% premium to a thirty-day average closing price, resulting in 267,531 shares
purchased at a price of $37.38 per Share. The 20% share premium is included as consideration for
the OGX-011 license and has been included in collaboration revenue.
In connection with the Collaboration Agreement and pursuant to the terms of agreements between the
Company and Isis relating to OGX-011, the Company will pay Isis $10 million which has been recorded
as research and development expense. The Company will also pay approximately $333,333 to the
University of British Columbia (UBC) pursuant to the terms of their license agreement relating to
OGX-011, which has been recorded as research and development expense. Pursuant to the terms of the
third-party agreements, the Company anticipates that it would be required to pay third-parties 31%
of any milestone payments that are not based on a percentage of net sales of the Licensed Product.
Pursuant to the terms of third-party agreements, the Company anticipates it will pay royalties to
third-parties of 4.88% to 8.00% of net sales, unless the Companys royalties are adjusted for
competition from generic compounds, in which case royalties to third-parties will also be subject
to adjustment on a country-by-country basis. Certain third-party royalties are tiered based on the
royalty rate received by the Company. Minimum royalty rates payable by the Company assume certain
third-party royalties are not paid at the time that the Licensed Product is marketed due to the
expiration of patents held by such third-parties. Maximum royalty rates assume all third-party
royalty rates currently in effect continue in effect at the time the Licensed Product is marketed.
Teva will receive the exclusive worldwide right and license to develop and commercialize products
containing OGX-011 and related compounds. The Company has an option to co-promote any Licensed
Product in the United States and Canada.
78
Teva is responsible for all costs relating to product commercialization including costs incurred in
relation to the Companys co-promotion option, except for start-up costs in advance of
commercialization.
Teva and the Company have developed a Clinical Development Plan under which three phase 3 clinical
trials will be initiated:
| |
|
|
a phase 3 clinical trial of the Licensed Product for second-line castrate resistant
prostate cancer, expected to initiate in the second quarter of 2010 subject to institutional review board approval and patient screening. The Company will have primary responsibility
for the oversight of this trial; |
| |
|
|
a phase 3 clinical trial of the Licensed Product for first-line castrate resistant
prostate cancer, expected to initiate in the third quarter of 2010; and |
| |
|
|
a phase 3 clinical trial of the Licensed Product for first-line NSCLC, expected to
initiate by early 2011. |
Teva will be responsible for conducting any other studies and development work necessary to obtain
required regulatory approvals. The Company may assume some of these activities if assigned by the
Joint Steering Committee. Teva will be responsible for all such costs. The Joint Steering Committee
will oversee the development and regulatory approval of any Licensed Product. The Company may
terminate its participation in the Joint Steering Committee at any time.
Funding responsibilities for the Clinical Development Plan will be allocated as follows:
| |
|
|
the Company will be required to spend $30 million in direct and indirect development
costs, and |
| |
|
|
Teva will fund all other expenses under the Clinical Development Plan. |
The Collaboration Agreement will remain in effect, on a country-by-country basis, until the
expiration of the obligation of Teva to pay royalties on sales of the Licensed Product in such
country (or earlier termination under its terms). Commencing after the completion of all three
phase 3 clinical trials set forth in the Clinical Development Plan, or upon early termination due
to a material adverse change in the Companys patent rights related to OGX-011 or safety issues or
futility as defined in the Collaboration Agreement, Teva may terminate the Collaboration
Agreement in its sole discretion upon three months notice if notice is given prior to regulatory
approval of a Licensed Product and upon six months notice if notice is given after such regulatory
approval. If Teva terminates the Collaboration for any reasons other than an adverse change in
OGX-011 patent rights, safety issues or futility determination as previously described, it will
remain responsible for paying for any remaining costs of all three phase 3 clinical trials, except
for Company Development Expenses. Either party may terminate the Collaboration Agreement for an
uncured material breach by the other party or upon the bankruptcy of either party. If the
Collaboration Agreement is terminated by the Company for other than an uncured material breach by
Teva, the Company will pay Teva a royalty on sales of Licensed Products. The percentage rates of
such royalties (which are in the single digits) depend if termination occurs prior to the first
regulatory approval in the United States or a primary European Market or after one of these
approvals. These royalties would expire on a country-by-country basis on the earlier of ten years
after the first commercial sale of a Licensed Product or certain thresholds related to generic
competition.
In the event of a change of control of the Company, within 90 days of the change of control, Teva
may terminate the joint steering committee in its sole discretion, terminate the co-promotion
option in its sole discretion if not then exercised by the Company or if exercised but not yet
executed by the Company, or terminate the co-promotion option if in its commercially reasonable
opinion co-promotion with the Companys successor would be materially detrimental to Tevas
interests.
79
Amendment to Isis and UBC License Agreements
To facilitate the execution and performance of the Collaboration Agreement, the Company and Isis
agreed to amend the Isis License Agreement and the Company and UBC agreed to amend the UBC License
Agreement, in each case, effective December 19 and December 20, 2009, respectively.
The amendment to the Isis License Agreement provides, among other things, that if the Company is
the subject of a change of control with a third party, where the surviving company immediately
following such change of control has the right to develop and sell the product, then (i) a
milestone payment of $20 million will be due and payable to Isis 21 days following the first
commercial sale of the product in the United States; and (ii) unless such surviving entity had
previously sublicensed the product and a royalty rate payable to Isis by the Company has been
established, the applicable royalty rate payable to Isis will thereafter be the maximum amount
payable under the Isis License Agreement. Any non-royalty milestone amounts previously paid will be
credited toward the $20 million milestone if not already paid. As a result of the $10 million
milestone payment payable to Isis in relation to the Collaboration Agreement, the remaining amount
owing in the event of change of control discussed above is a maximum of $10 million. As the Company
has now licensed the product to Teva and established a royalty rate payable to Isis, no royalty
rate adjustments would apply if Teva acquires the Company and is the surviving company. If the $30
million in advanced reimbursement of development activities has not been spent by OncoGenex prior
to the third anniversary of the Collaboration Agreement between OncoGenex and Teva, OncoGenex will
pay Isis an amount equal to 30% of any un-spent portion less $3.5 million.
5. REVERSE TAKEOVER
The consolidated financial statements account for the Arrangement between Sonus and OncoGenex
Technologies, whereby Sonus acquired all of the outstanding preferred shares, common shares and
convertible debentures of OncoGenex Technologies, as a reverse takeover wherein OncoGenex
Technologies is deemed to be the acquiring entity from an accounting perspective. The consolidated
results of operations of the Company include the results of operations of the combined company for
year ended December 31, 2009. The consolidated results of operations of the Company include the
results of operations of OncoGenex Technologies for the full year ended December 31, 2008 and the
results of OncoGenex Pharmaceuticals, Inc. following the completion of the Arrangement on August
21, 2008. The consolidated results of operations for year ended December 31, 2007 include only the
consolidated results of operations of OncoGenex Technologies and do not include historical results
of Sonus.
On August 12, 2008, OncoGenex Technologies shareholders approved the Arrangement described above
and on August 19, 2008, Sonus shareholders approved the Arrangement, an one-for-eighteen reverse
stock split of its common stock, and a reduction of Sonus authorized capital from 75,000,000
common shares to 11,019,930 common shares. The reverse stock split occurred immediately prior to
the closing of the Arrangement. Resulting fractional shares were eliminated. All information in the
financial statements and the notes thereto relating to the number of shares, price per share, and
per share amounts of common stock are presented on a post-split basis.
Under the purchase method of accounting, Sonus outstanding shares of common stock were valued
using the average closing price on Nasdaq of $5.04 for the two days prior through to the two days
subsequent to the announcement of the Arrangement on May 27, 2008. There were 2,059,898 shares of
common stock outstanding, as adjusted for the reverse stock split, on August 20, 2008, immediately
prior to closing. The fair value of the Sonus outstanding stock options were determined using the
Black-Scholes option pricing model with the following assumptions: stock price of $4.86, volatility
of 57.67% to 89.48%, risk-free interest rate of 1.73% to 3.89%, and expected lives ranging from
0.05 to 4.79 years. The fair value of the Sonus outstanding warrants were determined using the
Black-Scholes option pricing model with the following assumptions: stock price of $4.86, volatility
of 58.71%, risk-free interest rate 3.89%, and expected lives ranging from 0.99 to 1.08 years.
80
The final purchase price is summarized as follows (in thousands):
| |
|
|
|
|
Sonus common stock |
|
$ |
10,385 |
|
Fair value of options and warrants assumed |
|
|
71 |
|
Transaction costs of OncoGenex |
|
|
807 |
|
|
|
|
|
Total purchase price |
|
$ |
11,263 |
|
Under the purchase method of accounting, the total purchase price as shown in the table above is
allocated to the Sonus net tangible and identifiable intangible assets acquired and liabilities
assumed based on their fair values as of the date of the completion of the Arrangement. The final
purchase price allocation is as follows (in thousands):
| |
|
|
|
|
Cash |
|
$ |
5,464 |
|
Marketable securities |
|
|
14,808 |
|
Accounts receivable |
|
|
6 |
|
Interest receivable |
|
|
273 |
|
Other current assets |
|
|
175 |
|
Furniture and equipment |
|
|
1,186 |
|
Other long term assets |
|
|
497 |
|
Intangible assets |
|
|
280 |
|
Accounts payable |
|
|
(35 |
) |
Accrued expenses excluding severance payable |
|
|
(652 |
) |
Severance payable to employees as part of restructuring |
|
|
(1,322 |
) |
Severance payable to senior executives |
|
|
(1,440 |
) |
Excess facility loss |
|
|
(2,083 |
) |
Negative goodwill |
|
|
(5,894 |
) |
|
|
|
|
Total purchase price |
|
$ |
11,263 |
|
In accordance with SFAS 141, Business Combinations any excess of fair value of acquired net
assets over purchase price (negative goodwill) has been recognized as an extraordinary gain in the
period the Arrangement was completed. The excess has been allocated as a pro rata reduction of the
amounts that otherwise would have been assigned to the non-current acquired assets. Prior to
allocation of the excess negative goodwill OncoGenex has reassessed whether all acquired assets and
assumed liabilities have been identified and recognized and performed remeasurements to verify that
the consideration paid, assets acquired, and liabilities assumed have been properly valued. The
remaining excess has been recognized as an extraordinary gain. There was no other impact to other
comprehensive income.
The final pro rata reduction of non-current and intangible assets acquired is as follows (in
thousands):
| |
|
|
|
|
Negative goodwill |
|
$ |
(5,894 |
) |
|
|
|
|
Furniture and equipment |
|
|
1,186 |
|
Intangible assets |
|
|
280 |
|
|
|
|
|
Excess negative goodwill |
|
$ |
(4,428 |
) |
81
Pro Forma Results of Operations
The results of operations the combined Company are reflected in the consolidated financial
statements from the date of the completion of the Arrangement on August 21, 2008. The following
table presents pro forma results of operations and gives effect to the business combination
transaction as if it were consummated at the beginning of the period presented. The pro forma
results of operations are not necessarily indicative of what would have occurred had the business
combination been completed at the beginning of the retrospective periods or of the results that may
occur in the future.
| |
|
|
|
|
| |
|
For the year ended |
|
| |
|
December 31, |
|
| (in thousands except shareholder and per share amounts) |
|
2008 |
|
Revenue |
|
|
|
|
Net loss applicable to common shareholders |
|
$ |
(28,102 |
) |
Net loss per share basic and diluted |
|
$ |
(15.36 |
) |
Weighted average shares |
|
|
1,829,276 |
|
6. FAIR VALUE MEASUREMENTS
With the adoption of ASC 820 Fair Value Measurements and Disclosures, beginning January 1, 2008,
assets and liabilities recorded at fair value in the balance sheets are categorized based upon the
level of judgment associated with the inputs used to measure their fair value. For certain of the
Companys financial instruments including cash and cash equivalents, amounts receivable, and
accounts payable the carrying values approximate fair value due to their short-term nature.
ASC 820 specifies a hierarchy of valuation techniques based on whether the inputs to those
valuation techniques are observable or unobservable. In accordance with ASC 820, these inputs are
summarized in the three broad level listed below:
| |
|
|
Level 1 Quoted prices in active markets for identical securities; |
| |
|
|
Level 2 Other significant observable inputs that are observable through corroboration
with market data (including quoted prices in active markets for similar securities); |
| |
|
|
Level 3 Significant unobservable inputs that reflect managements best estimate of
what market participants would use in pricing the asset or liability. |
As quoted prices in active markets are not readily available, the Company obtains estimates for the
fair value of financial instruments through independent pricing service providers.
In determining the appropriate levels, the Company performed a detailed analysis of the assets and
liabilities that are subject to ASC 820.
The Company invests its excess cash in accordance with investment guidelines that limit the credit
exposure to any one financial institution other than securities issued by the U.S. Government. The
guidelines also specify that the financial instruments be issued by institutions with strong credit
ratings. These securities are generally not collateralized and mature within one year.
A description of the valuation techniques applied to the Companys marketable securities measured
at fair value on a recurring basis follows.
Financial Instruments
Cash
Significant amounts of cash are held on deposit with a large well established Canadian financial
institution.
U.S. Government and Agency Securities
U.S. Government Securities U.S. government securities are valued using quoted market
prices. Valuation adjustments are not applied. Accordingly, U.S. government securities are
categorized in Level 1 of the fair value hierarchy.
82
U.S. Agency Securities U.S. agency securities are comprised of two main categories
consisting of callable and non-callable agency issued debt securities. Non-callable agency issued
debt securities are generally valued using quoted market prices. Callable agency issued debt
securities are valued by benchmarking model-derived prices to quoted market prices and trade data
for identical or comparable securities. Actively traded non-callable agency issued debt securities
are categorized in Level 1 of the fair value hierarchy. Callable agency issued debt securities are
categorized in Level 2 of the fair value hierarchy.
Corporate and Other Debt
Corporate Bonds and Commercial Paper The fair value of corporate bonds and commercial
paper is estimated using recently executed transactions, market price quotations (where
observable), bond spreads or credit default swap spreads adjusted for any basis difference between
cash and derivative instruments. The spread data used are for the same maturity as the bond. If the
spread data does not reference the issuer, then data that reference a comparable issuer are used.
When observable price quotations are not available, fair value is determined based on cash flow
models with yield curves, bond or single name credit default swap spreads and recovery rates based
on collateral values as significant inputs. Corporate bonds and commercial paper are generally
categorized in Level 2 of the fair value hierarchy; in instances where prices, spreads or any of
the other aforementioned key inputs are unobservable, they are categorized in Level 3 of the
hierarchy.
The following table presents information about our assets and liabilities that are measured at fair
value on a recurring basis as of December 31, 2009, and indicates the fair value hierarchy of the
valuation techniques we utilized to determine such fair value:
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| (in thousands) |
|
Level 1 |
|
|
Level 2 |
|
|
Level 3 |
|
|
2009 |
|
Marketable Securities |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Money market securities |
|
$ |
5,941 |
|
|
$ |
|
|
|
$ |
|
|
|
$ |
5,941 |
|
U.S. treasury securities |
|
|
6,033 |
|
|
|
|
|
|
|
|
|
|
|
6,033 |
|
Other government debt securities |
|
|
401 |
|
|
|
1,103 |
|
|
|
|
|
|
|
1,504 |
|
Corporate bonds and commercial
paper |
|
|
|
|
|
|
3,500 |
|
|
|
|
|
|
|
3,500 |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$ |
12,375 |
|
|
$ |
4,603 |
|
|
$ |
|
|
|
$ |
16,978 |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| (in thousands) |
|
Level 1 |
|
|
Level 2 |
|
|
Level 3 |
|
|
2008 |
|
Marketable Securities |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Money market securities |
|
$ |
4,667 |
|
|
$ |
|
|
|
$ |
|
|
|
$ |
4,667 |
|
Corporate bonds and commercial
paper |
|
|
|
|
|
|
5,594 |
|
|
|
|
|
|
|
5,594 |
|
Other government debt securities |
|
|
|
|
|
|
1,009 |
|
|
|
|
|
|
|
1,009 |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$ |
4,667 |
|
|
$ |
6,603 |
|
|
$ |
|
|
|
$ |
11,270 |
|
83
Marketable securities consist of the following:
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
Gross |
|
|
Gross |
|
|
|
|
| |
|
Amortized |
|
|
Unrealized |
|
|
Unrealized |
|
|
Estimated |
|
| (in thousands) |
|
Cost |
|
|
Gain |
|
|
Loss |
|
|
Fair Value |
|
2009 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Money market securities |
|
$ |
5,941 |
|
|
$ |
|
|
|
$ |
|
|
|
$ |
5,941 |
|
U.S. treasury securities |
|
|
6,033 |
|
|
|
1 |
|
|
|
(2 |
) |
|
|
6,032 |
|
Other government debt securities |
|
|
1,504 |
|
|
|
1 |
|
|
|
|
|
|
|
1,505 |
|
Corporate bonds and commercial
paper |
|
|
3,500 |
|
|
|
|
|
|
|
|
|
|
|
3,500 |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$ |
16,978 |
|
|
$ |
2 |
|
|
$ |
(2 |
) |
|
$ |
16,978 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
2008 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Money market securities |
|
$ |
4,667 |
|
|
$ |
|
|
|
$ |
|
|
|
$ |
4,667 |
|
Corporate bonds and commercial
paper |
|
|
5,599 |
|
|
|
2 |
|
|
|
(7 |
) |
|
|
5,594 |
|
Other government debt securities |
|
|
1,007 |
|
|
|
2 |
|
|
|
|
|
|
|
1,009 |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$ |
11,273 |
|
|
$ |
4 |
|
|
$ |
(7 |
) |
|
$ |
11,270 |
|
At December 31, 2009 $5,941,000 of money market securities, $5,019,000 of Government debt
securities and $3,500,000 of Commercial paper in the above tables are included in cash equivalents
as the securities have maturities of 90 days or less at the time of purchase. At December 31, 2008
$4,667,000 of money market securities, $1,802,000 of Corporate bonds in the above tables are
included in cash equivalents as the securities have maturities of 90 days or less at the time of
purchase. The remaining securities all mature within one year of the balance sheet date and are
included in short-term investments.
There were no significant realized or unrealized gains or losses on the sales of marketable
securities in the periods ended December 31, 2009 or December 31, 2008, and no significant
unrealized gains or losses are included in accumulated other comprehensive income as at December
31, 2009. Realized gains and losses are transferred out of accumulated other comprehensive income
into interest income using the specific identification method.
All of the marketable securities held as of December 31, 2009 had maturities of one year or less.
The Company only invests in A (or equivalent) rated securities with maturities of one year or less.
The Company does not believe that there are any other than temporary impairments related to its
investment in marketable securities at December 31, 2009 given the quality of the investment
portfolio, its short-term nature, and subsequent proceeds collected on sale of securities that
reached maturity.
7. PROPERTY AND EQUIPMENT
| |
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
Accumulated |
|
|
Net Book |
|
| (In thousands) |
|
Cost |
|
|
Amortization |
|
|
Value |
|
| |
|
$ |
|
|
$ |
|
|
$ |
|
December 31, 2009 |
|
|
|
|
|
|
|
|
|
|
|
|
Computer equipment |
|
|
276 |
|
|
|
271 |
|
|
|
5 |
|
Furniture and fixtures |
|
|
113 |
|
|
|
90 |
|
|
|
23 |
|
Leasehold improvements |
|
|
42 |
|
|
|
42 |
|
|
|
|
|
Equipment under capital lease |
|
|
66 |
|
|
|
22 |
|
|
|
44 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
497 |
|
|
|
425 |
|
|
|
72 |
|
December 31, 2008 |
|
|
|
|
|
|
|
|
|
|
|
|
Computer equipment |
|
|
268 |
|
|
|
248 |
|
|
|
20 |
|
Furniture and fixtures |
|
|
94 |
|
|
|
70 |
|
|
|
23 |
|
Leasehold improvements |
|
|
38 |
|
|
|
38 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
400 |
|
|
|
356 |
|
|
|
44 |
|
84
8. SEVERANCE CHARGES AND OTHER RESTRUCTURING ACTIVITIES
On August 21, 2008, immediately following the completion of the Arrangement (Note 5), the Company
reduced its workforce by approximately 49%. Severance payable at the date of the restructuring in
connection with former employees of Sonus was $1,322,000 and has been accounted for in accordance
with EITF No. 95-3, Recognition of Liabilities in Connection with a Purchase Business Combination
as part of the purchase price allocation (Note 5). During 2008 the Company made payments totalling
$1,186,000 and the amount owing at December 31, 2008 was $137,000. All remaining severance
liabilities relating to transaction-related workforce reductions were paid out during 2009, and the
amount owing at December 31, 2009 was nil.
Prior to the Arrangement, Sonus entered into a non-cancellable lease arrangement for office space
located in Bothell, Washington, which is considered to be in excess of the Companys current
requirements. The Company is currently in the process of evaluating opportunities to exit or sublet
portions of the leased space and recorded an initial restructuring charge of $2,084,000 on August
21, 2008 as part of the purchase price allocation (Note 5). The liability is computed as the
present value of the difference between the remaining lease payments due less the estimate of net
sublease income and expenses and has been accounted for in accordance with EITF No. 95-3,
Recognition of Liabilities in Connection with a Purchase Business Combination. This represents
the Companys best estimate of the fair value of the liability. Subsequent changes in the liability
due to accretion, or changes in estimates of sublease assumptions will be recognized as adjustments
to restructuring charges in future periods. During 2008, $362,000 was amortized into income,
resulting in a remaining liability at December 31, 2008 of $1,722,000.
In June 2009 the Company revised its sublease income assumptions used to estimate the fair value of
the excess lease facility liability. These assumptions were subsequently revised again in December
2009. These changes in estimate resulted in increases in the fair value of the excess lease
liability and $494,000 and $3,457,000 in charges to research and development expense recorded in
June 2009 and December 2009, respectively, to reflect these changes in estimate. These changes in
estimate had a $0.69 impact on loss per common share for year ended December 31, 2009. The
estimated carrying value of the liability remaining at December 31, 2009 with respect to excess
facilities is $4,645,000.
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
Remaining |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Remaining |
|
| |
|
Liability at |
|
|
|
|
|
|
Amortization |
|
|
Additional |
|
|
Liability at |
|
| |
|
December 31, |
|
|
Payments |
|
|
of excess |
|
|
Liability |
|
|
December 31, |
|
| (in thousands) |
|
2008 |
|
|
made |
|
|
lease facility |
|
|
Recorded |
|
|
2009 |
|
Employee severance |
|
$ |
137 |
|
|
$ |
137 |
|
|
|
|
|
|
|
|
|
|
|
0 |
|
Current portion of excess lease facility |
|
$ |
632 |
|
|
|
|
|
|
|
(884 |
) |
|
$ |
1,549 |
|
|
$ |
1,297 |
|
Long-term portion of excess lease
facility |
|
$ |
1,090 |
|
|
|
|
|
|
|
(144 |
) |
|
$ |
2,402 |
|
|
$ |
3,348 |
|
9. OTHER ASSETS
Other assets include deposits paid for office space in accordance with the terms of the operating
lease agreements.
10. INCOME TAX
[a] The reconciliation of income tax attributable to operations computed at the statutory tax
rate to income tax expense, using a statutory tax rate of 34% for the years ended December 31, 2009
and December 31, 2008, and 34.12% for the year ended December 31, 2007 is as follows. 2007 balances
have been derived from the audited financial statements of OncoGenex Technologies Inc., a Canadian
corporation (OncoGenex Technologies), which is subject to combined Canadian federal and
provincial statutory tax rates for December 31, 2009, 2008, and 2007 of 30%, 31%, and 34.12%,
respectively. Following the reverse takeover by OncoGenex Technologies of Sonus Pharmaceuticals,
Inc. (which subsequently changed its name to OncoGenex Pharmaceuticals, Inc.) in 2008, OncoGenex
Technologies became a wholly owned subsidiary of the Company, which is a Delaware incorporated
company subject to US Federal Statutory rates of 34% for all three years presented.
85
For the purposes of estimating the tax rate in effect at the time that deferred tax assets and
liabilities are expected to reverse, management uses the furthest out available future tax rate in
the applicable jurisdictions. For the years ended December 31, 2009, 2008, and 2007 the future
Canadian enacted rates we used were 25%, 26%, and 27%, respectively, while for the US the future
enacted rate we used was 34% for all three periods presented.
| |
|
|
|
|
|
|
|
|
|
|
|
|
| (In thousands) |
|
2009 |
|
|
2008 |
|
|
2007 |
|
Income taxes at statutory rates |
|
|
(838 |
) |
|
|
(3,635 |
) |
|
|
(2,669 |
) |
Expenses not deducted for tax purposes(1) |
|
|
151 |
|
|
|
3,991 |
|
|
|
389 |
|
Effect of tax rate changes on deferred tax assets and liabilities |
|
|
|
|
|
|
|
|
|
|
|
|
Effect of foreign tax (Canadian) rate changes on deferred tax assets and liabilities |
|
|
(130 |
) |
|
|
974 |
|
|
|
1,721 |
|
Reduction in benefit of operating losses |
|
|
542 |
|
|
|
339 |
|
|
|
|
|
Reduction in the benefit of other tax attributes |
|
|
367 |
|
|
|
|
|
|
|
|
|
Impact of Withholding Tax |
|
|
3,000 |
|
|
|
|
|
|
|
|
|
Foreign exchange effect on valuation allowance |
|
|
(1,437 |
) |
|
|
2,244 |
|
|
|
(1,643 |
) |
Investment tax credits |
|
|
(180 |
) |
|
|
(180 |
) |
|
|
|
|
Research and development tax credits |
|
|
(32 |
) |
|
|
|
|
|
|
(96 |
) |
Change in valuation allowance |
|
|
1,628 |
|
|
|
1,643 |
|
|
|
2,685 |
|
Reversal of tax effect of income of Sonus prior to the Arrangement (2) |
|
|
|
|
|
|
(5,310 |
) |
|
|
|
|
Canadian Part VI.1 tax on redeemable convertible preferred shares (3) |
|
|
|
|
|
|
(2,125 |
) |
|
|
676 |
|
Part VI.I tax deduction |
|
|
|
|
|
|
|
|
|
|
(519 |
) |
Book to tax return adjustments |
|
|
(60 |
) |
|
|
|
|
|
|
|
|
Other |
|
|
|
|
|
|
|
|
|
|
169 |
|
|
|
|
|
|
|
|
|
|
|
Income tax expense |
|
|
3,011 |
|
|
|
(2,059 |
) |
|
|
713 |
|
| |
|
|
| (1) |
|
The increase in 2008 from 2007 was primarily attributable to amounts recognized in connection
with the reverse takeover by OncoGenex Technologies Inc. of Sonus Pharmaceuticals, Inc. (which
subsequently changed its name to OncoGenex Pharmaceuticals, Inc.) (Sonus) during 2008 (the
Arrangement), including facility-related charges and the conversion of redeemable convertible
preferred shares into equity following the Arrangement. |
| |
| (2) |
|
This line item represents the adjustment required to the 2008 tax provision for amounts
associated with Sonus during the portion of the year prior to the Arrangement. The amount of $5,310
represents the pre-arrangement loss of Sonus of $15,617 multiplied by the US Federal Statutory tax
rate of 34%. Offsetting the $5,310 was $5,310 included in line item Change in valuation
allowance. |
| |
| (3) |
|
Canadian Part VI.1 tax on redeemable convertible preferred shares was accrued during each year
such shares were outstanding. This tax would have been payable upon retraction of redeemable
convertible preferred shares. These shares would have been retractable at any time after August
10. 2010. In 2008, subsequent to the completion of the Arrangement, all convertible redeemable
preferred shares were converted into equity and as a result the associated aggregate tax liability
was reversed. |
[b] At December 31, 2009, the Company has investment tax credits of $509,000 (2008$268,000)
available to reduce future Canadian income taxes otherwise payable. The Company also has
non-capital loss carry forwards for financial statement purposes of $21,057,000 (2008$25,623,000)
available to offset future taxable income in Canada and federal net operating loss carryforwards of
$127,424,000 (2008- 124,298,000) to offset future taxable income in the United States.
The initial public offering of common stock by the Company in 1995 caused an ownership change
pursuant to applicable regulations in effect under the Internal Revenue Code of 1986. Therefore,
the Companys use of losses incurred through the date of ownership change will be limited during
the carryforward period and may result in the expiration of net operating loss carryforwards in the
United States before utilization.
86
The investment tax credits and non-capital losses and net operating losses for income tax purposes
expire as follows (in thousands):
| |
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
Non-capital |
|
| |
|
|
|
|
|
and Net |
|
| |
|
Investment |
|
|
Operating |
|
| |
|
Tax Credits |
|
|
Losses |
|
| |
|
$ |
|
|
$ |
|
2010 |
|
|
|
|
|
|
5,702 |
|
2011 |
|
|
|
|
|
|
47 |
|
2012 |
|
|
|
|
|
|
44 |
|
2013 |
|
|
|
|
|
|
10,795 |
|
2014 |
|
|
|
|
|
|
1,624 |
|
2015 |
|
|
|
|
|
|
|
|
2016 |
|
|
|
|
|
|
|
|
2017 |
|
|
|
|
|
|
|
|
2018 |
|
|
7 |
|
|
|
|
|
2019 |
|
|
64 |
|
|
|
32 |
|
2020 |
|
|
|
|
|
|
2,745 |
|
2021 |
|
|
2 |
|
|
|
400 |
|
2022 |
|
|
2 |
|
|
|
11,766 |
|
2023 |
|
|
1 |
|
|
|
10,785 |
|
2024 |
|
|
|
|
|
|
16,814 |
|
2025 |
|
|
232 |
|
|
|
9,101 |
|
2026 |
|
|
67 |
|
|
|
31,713 |
|
2027 |
|
|
|
|
|
|
27,440 |
|
2028 |
|
|
23 |
|
|
|
14,938 |
|
2029 |
|
|
111 |
|
|
|
4,535 |
|
|
|
|
|
|
|
|
|
|
|
509 |
|
|
|
148,481 |
|
In addition, the Company has unclaimed tax deductions of approximately $8,484,000 related to
scientific research and experimental development expenditures available to carry forward
indefinitely to reduce Canadian taxable income of future years. The Company also has research and
development tax credits of $1,682,000 available to reduce future taxes payable in the United
States. The research and development tax credits expire between 2010 and 2029.
[c] Significant components of the Companys deferred tax assets as of December 31 are shown
below (in thousands):
| |
|
|
|
|
|
|
|
|
| |
|
2009 |
|
|
2008 |
|
| |
|
$ |
|
|
$ |
|
Deferred tax assets: |
|
|
|
|
|
|
|
|
Tax basis in excess of book value of assets |
|
|
1,202 |
|
|
|
692 |
|
Non-capital loss carryforwards |
|
|
48,588 |
|
|
|
48,923 |
|
Research and development deductions and
credits |
|
|
4,210 |
|
|
|
3,777 |
|
Part VI.1 tax deduction |
|
|
|
|
|
|
|
|
Share issue costs |
|
|
119 |
|
|
|
144 |
|
Stock options |
|
|
1,689 |
|
|
|
1,560 |
|
Capital loss carryforward |
|
|
51 |
|
|
|
268 |
|
Deferred rent |
|
|
1,697 |
|
|
|
494 |
|
Foreign tax credit |
|
|
3,273 |
|
|
|
|
|
Other |
|
|
27 |
|
|
|
|
|
|
|
|
|
|
|
|
Total deferred tax assets |
|
|
60,856 |
|
|
|
55,858 |
|
Valuation allowance |
|
|
(60,856 |
) |
|
|
(55,858 |
) |
87
The potential income tax benefits relating to these deferred tax assets have not been recognized in
the accounts as their realization did not meet the requirements of more likely than not under the
liability method of tax allocation. Accordingly, a valuation allowance has been recorded and no
deferred tax assets have been recognized as at December 31, 2009 and 2008. The change in valuation
allowance is due to a decrease in deferred tax assets from OncoGenex Technologies Inc. and the
addition of deferred tax assets from OncoGenex Pharmaceuticals, Inc.
[d] Under FIN 48, the benefit of an uncertain tax position that is more likely than not of being
sustained upon audit by the relevant taxing authority must be recognized at the largest amount that
is more likely than not to be sustained. No portion of the benefit of an uncertain tax position may
be recognized if the position has less than a 50% likelihood of being sustained.
A reconciliation of the unrecognized tax benefits of uncertain tax positions for the year ended
December 31, 2009 is as follows:
| |
|
|
|
|
| (in thousands) |
|
$ |
|
Balance as of December 31, 2007 |
|
|
614 |
|
Additions based on the reverse takeover of Sonus |
|
|
1,355 |
|
Additions based on tax positions related to the current year |
|
|
342 |
|
Deductions based on tax positions related to the current year |
|
|
(355 |
) |
|
|
|
|
Balance as of December 31, 2008 |
|
|
1,956 |
|
Additions based on tax positions related to the current year |
|
|
143 |
|
Deductions based on tax positions related to the current year |
|
|
(90 |
) |
|
|
|
|
Balance as of December 31, 2009 |
|
|
2,009 |
|
As of December 31, 2009, unrecognized benefits of approximately $2,009,000, if recognized, would
affect the Companys effective tax rate.
The Companys accounting policy is to treat interest and penalties relating to unrecognized tax
benefits as a component of income taxes. As of December 31, 2009 and December 31, 2008 the Company
had no accrued interest and penalties related to income taxes.
The Company is subject to taxes in Canada and the U.S. until the applicable statute of limitations
expires. Tax audits by their very nature are often complex and can require several years to
complete.
| |
|
|
|
|
| Tax |
|
Years open to |
|
| Jurisdiction |
|
examination |
|
Canada |
|
|
2003 to 2009 |
|
US |
|
|
2003 to 2009 |
|
11. COMMON STOCK
[a] Authorized
11,019,930 authorized common voting shares, par value of $0.001
88
[b] Issued and outstanding shares
As at August 20, 2008, there were 118,801 common shares of OncoGenex Technologies (on a
post-conversion basis) and 2,059,898 shares of common stock of Sonus outstanding. As part of the
arrangement (note 5), Sonus agreed to issue 3,449,393 shares of common stock, after accounting for
the elimination of resulting fractional shares, in exchange for all the common shares, preferred
shares and convertible debentures of OncoGenex Technologies. As a result, all common shares of
OncoGenex Technologies are now held by the Company and have been eliminated on consolidation.
During the year ended December 31, 2009, the Company issued 37,388 common shares upon exercise of
stock options (year ended December 31, 2008 34,601, year ended December 31, 2007 nil). The
Company issues new shares to satisfy stock option exercises.
Escrow shares
As part of the Arrangement (note 5), 1,388,875 of the shares of common stock issuable to the
holders of OncoGenex Technologies securities were placed into escrow at the closing of the
Arrangement and were to be released from escrow upon the achievement of certain agreed-upon
milestones relating to OncoGenex product candidates OGX-011, OGX-427 and OGX-225 and the future
price of our common stock. The milestone shares were issued and placed into escrow at the closing
of the Arrangement.
On July 24, 2008, the Company announced the completion of a Special Protocol Assessment on the
patient population, trial design, trial endpoints, statistical analyses and size of a registration
clinical trial with OGX-011. The achievement of this milestone resulted in the release of 25%
(347,237) of the shares held in escrow.
On October 7, 2008 the Company concluded a meeting with the U.S. Food and Drug Administration
(FDA), at which the FDA agreed that durable pain palliation is an acceptable and desirable trial
endpoint to support product marketing approval for OGX-011 as a treatment for CRPC. In addition,
OncoGenex reported that the FDA provided guidance on the submitted protocol including
recommendations on trial endpoints, the appropriate patient population, entry criteria and trial
conduct. Based on the results of this meeting, the Board of Directors of OncoGenex approved the
release of 25% (347,207) of the shares held in escrow. The escrow agreements provided for the
release of 25% of the shares held in escrow following the occurrence of a meeting with the FDA to
confirm that pain palliation is an appropriate primary endpoint to support a product marketing
approval in prostate cancer.
On December 3, 2008, the Company announced positive survival results from a randomized phase 2
clinical trial of OGX-011 in combination with docetaxel and prednisone (the OGX-011 arm) compared
to docetaxel and prednisone alone (the control arm) for first-line treatment of metastatic
castrate resistant prostate cancer. The OGX-011 arm demonstrated a 10.6 month median overall
survival advantage over the median survival observed in the control arm. The escrow agreements
provided for the release of 50% (694,431) of the original number of shares held in escrow following
the demonstration of at least a two-month improvement in survival in the OGX-011 arm as compared to
the control arm. Based on the median overall survival advantage of the OGX-011 arm, the Board of
Directors of OncoGenex Pharmaceuticals approved the release of all of the remaining shares held in
escrow pursuant to agreements related to the Arrangement.
As at December 31, 2008 all milestone shares had been released from escrow.
July 2009 Financing
On July 24, 2009, the Company completed a registered direct offering with certain institutional
investors covering the sale of 475,000 shares of common stock at a price of $20 per share under a
shelf registration statement on Form S-3 (No. 333-160251) that was declared effective by the SEC on
July 17, 2009. The transaction provided net proceeds of approximately $9.3 million after deducting
costs associated with the offering.
89
2009 Teva Share Purchase Agreement
On December 20, 2009 the Company and Teva also entered into a stock purchase agreement (the Stock
Purchase Agreement). Pursuant to the terms of the Stock Purchase Agreement, Teva made a $10
million equity investment in the Company through its purchase of 267,531 shares of the common
shares at a price of $37.38 per Share, which was a 20% premium to a thirty-day average closing
price prior to the announcement. The transaction provided net proceeds of approximately $9,970,000
after deducting costs associated with the offering. The 20% share premium has been allocated to
revenue, resulting in a net amount of $7,930,000 included in equity.
[c] Stock options
OncoGenex Technologies Inc. Stock Option Plan
In September 2003, the Board of Directors of OncoGenex Technologies approved an amended stock
option plan (the OncoGenex Technologies Plan), which was an amendment of the stock option plan
first established in October 2001. The OncoGenex Technologies Plan was subsequently approved by
shareholders on August 12, 2008. Under this plan, the Company may grant options to purchase common
shares in the Company to employees, directors, officers, and consultants of the Company. The
exercise price of the options is determined by the Board but generally will be at least equal to
the fair value of the common shares at the grant date.
The options vest in accordance with terms as determined by the Board, typically over three or four
years for options issued to employees. The expiry date for each option is set by the Board with a
maximum expiry date of seven years and a minimum expiry of five years from the date of grant.
On August 21, 2008, under the Arrangement (note 5), each option to purchase shares of OncoGenex
Technologies common stock (OncoGenex Technologies Option) was exchanged for an option to purchase
shares of OncoGenex common stock. Specifically, each OncoGenex Technologies Option was exchanged
for an option to purchase the amount of shares of common stock of OncoGenex equal to the product of
(a) the share exchange ratio of the Arrangement (Share Exchange Ratio), as adjusted by the
one-for-eighteen reverse stock split, (b) multiplied by the number of OncoGenex Technologies shares
of common stock subject to each OncoGenex Technologies Option. The exercise price of each OncoGenex
Technologies Option was also adjusted to an amount equal to the product of (x) the exercise price
per share of each OncoGenex Technologies Option immediately prior to the effective time of the
arrangement, (y) divided by the Share Exchange Ratio, as adjusted by the one-for-eighteen reverse
stock split, (z) multiplied by the noon buying rate of exchange for one U.S. dollar in Canadian
dollars as published by the Federal Reserve Bank of New York on the date immediately prior to the
Arrangement.
Sonus Option Plans
Prior to the Arrangement, Sonus had options outstanding under a number of share option plans that
had been approved by shareholders as follows: (a) the Incentive Stock Option, Nonqualified Stock
Option and Restricted Stock Purchase Plan 1991 (1991 Plan), (b) the 1999 Nonqualified Stock
Incentive Plan (1999 Plan), (c) the 2000 Stock Incentive Plan (2000 Plan), and (d) the 2007
Performance Incentive Plan (2007 Plan) (collectively referred to as the Sonus Plans).
Pursuant to certain change of control provisions in the 1999 Plan and the 2000 Plan, all
outstanding options granted under those plans were cancelled immediately prior to the Arrangement.
Pursuant to the change of control provision in the 2007 Plan, vesting of options granted under the
2007 Plan was accelerated and all outstanding options granted under that plan became fully vested
immediately prior to the Arrangement. No changes were made to the 1991 Plan. All outstanding
options issued under the 1991 Plan were fully vested prior to the Arrangement.
All outstanding options to purchase common shares under the Sonus Plans have been adjusted to
reflect the one-for-eighteen reverse stock split. Because this modification was designed to
equalize the fair value of an award before and after an equity restructuring, no incremental
compensation cost is recognized.
90
ASC 718 Compensation Stock Compensation
The Company recognizes expense related to the fair value of our stock-based compensation awards
using the provisions of ASC 718. The Company uses the Black-Scholes option pricing model as the
most appropriate fair value method for its awards and recognizes compensation expense for stock
options on a straight-line basis over the requisite service period. In valuing its options using
the Black-Scholes option pricing model, the Company makes assumptions about risk-free interest
rates, dividend yields, volatility and weighted average expected lives, including estimated
forfeiture rates of the options.
The expected life was calculated based on the simplified method as permitted by the SECs Staff
Accounting Bulletin 110, Share-Based Payment. The Company considers the use of the simplified
method appropriate because of the lack of sufficient historical exercise data following the reverse
takeover of Sonus. The computation of expected volatility was based on the historical volatility of
comparable companies from a representative peer group selected based on industry and market
capitalization. The risk-free interest rate was based on a U.S. Treasury instrument whose term is
consistent with the expected life of the stock options. In addition to the assumptions above, as
required under ASC 718, management made an estimate of expected forfeitures and is recognizing
compensation costs only for those equity awards expected to vest. Forfeiture rates are estimated
using historical actual forfeiture rate that resulted over the estimated life of the option grant
for options granted as of the beginning of the forfeiture measurement period. These rates are
adjusted on a quarterly basis and any change in compensation expense is recognized in the period of
the change. The Company has never paid or declared dividends on our common stock and do not expect
to pay cash dividends in the foreseeable future.
The estimated fair value of stock options granted in the respective periods was determined using
the Black-Scholes option pricing model using the following weighted average assumptions:
| |
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
Years ended |
|
| |
|
December 31, |
|
| |
|
2009 |
|
|
2008 |
|
|
2007 |
|
Risk-free interest rates |
|
|
2.85 |
% |
|
|
1.71 |
% |
|
|
4.63 |
% |
Expected dividend yield |
|
|
0 |
% |
|
|
0 |
% |
|
|
0 |
% |
Expected life |
|
6.1 years |
|
|
5.2 years |
|
|
5 years |
|
Expected volatility |
|
|
74 |
% |
|
|
77 |
% |
|
|
100 |
% |
The weighted average fair value of stock options granted during the year ended December 31, 2009
was $12.03 per share (December 31, 2008 $1.86 and December 31, 2007 $14.50).
Total stock-based compensation expense included in the Companys statements of operations for the
years ended December 31, 2009, 2008 and 2007 was $380,000, $174,000 and $263,000 respectively.
The results for the periods set forth below included share-based compensation expense in the
following expense categories of the consolidated statements of operations:
| |
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
Years ended |
|
| |
|
December 31, |
|
| (in thousands) |
|
2009 |
|
|
2008 |
|
|
2007 |
|
| |
|
$ |
|
|
$ |
|
|
$ |
|
Research and development |
|
|
88 |
|
|
|
65 |
|
|
|
93 |
|
General and administrative |
|
|
292 |
|
|
|
109 |
|
|
|
170 |
|
|
|
|
|
|
|
|
|
|
|
Total share-based compensation |
|
|
380 |
|
|
|
174 |
|
|
|
263 |
|
|
|
|
|
|
|
|
|
|
|
91
Options vest in accordance with terms as determined by the Board, typically over three or four
years for employee grants and one year for Board of Director option grants. The expiry date for
each option is set by the Board with, which is typically seven to ten years. The exercise price of
the options is determined by the Board but generally will be at least equal to the fair value of
the share at the grant date. As at December 31, 2009 the Company has reserved 856,555 common shares
for issuance of stock options to employees, directors, officers and consultants of the Company,
under its various equity compensation plans, of which 53,684 (December 31, 2008 170,911) are
available for future issuance.
Stock option transactions and the number of stock options outstanding, after giving effect to the
adjustments made to the OncoGenex Technologies Plan options and Sonus Plans options described
above, are summarized below:
| |
|
|
|
|
|
|
|
|
| |
|
Number |
|
|
|
|
| |
|
of |
|
|
Weighted |
|
| |
|
Optioned |
|
|
Average |
|
| |
|
Common |
|
|
Exercise |
|
| |
|
Shares |
|
|
Price |
|
| |
|
# |
|
|
$ |
|
Balance, December 31, 2006 |
|
|
309,919 |
|
|
|
3.95 |
|
Option grants |
|
|
15,984 |
|
|
|
18.93 |
|
Option cancellations |
|
|
(1,672 |
) |
|
|
18.93 |
|
|
|
|
|
|
|
|
Balance, December 31, 2007 |
|
|
324,231 |
|
|
|
4.61 |
|
Additions from Sonus Option Plans |
|
|
98,249 |
|
|
|
27.89 |
|
Option grants |
|
|
397,150 |
|
|
|
2.93 |
|
Option exercises |
|
|
(34,601 |
) |
|
|
3.57 |
|
Option cancellations |
|
|
(61,886 |
) |
|
|
28.08 |
|
|
|
|
|
|
|
|
Balance, December 31, 2008 |
|
|
723,143 |
|
|
|
4.88 |
|
Option grants |
|
|
130,400 |
|
|
|
17.52 |
|
Option cancellations |
|
|
(8,154 |
) |
|
|
7.04 |
|
Option exercises |
|
|
(37,388 |
) |
|
|
3.79 |
|
Option forfeited |
|
|
(5,130 |
) |
|
|
6.42 |
|
|
|
|
|
|
|
|
Balance, December 31, 2009 |
|
|
802,871 |
|
|
|
6.95 |
|
92
The following table summarizes information about stock options outstanding at December 31, 2009:
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
Weighted- |
|
|
|
|
|
|
|
|
|
|
Weighted- |
|
| |
|
|
|
Options |
|
|
Average |
|
|
Options |
|
|
|
|
|
|
Average |
|
| |
|
|
|
Outstanding |
|
|
Remaining |
|
|
Exercisable |
|
|
Weighted- |
|
|
Remaining |
|
| |
|
|
|
Number of |
|
|
Contractual |
|
|
Number of |
|
|
Average |
|
|
Contractual |
|
| |
|
|
|
Shares |
|
|
Life |
|
|
Shares |
|
|
Exercise |
|
|
Life |
|
| Exercise Prices |
|
|
Outstanding |
|
|
(in years) |
|
|
Outstanding |
|
|
Price |
|
|
(in years) |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
2.69 |
|
|
|
68,000 |
|
|
|
5.8 |
|
|
|
68,000 |
|
|
|
2.69 |
|
|
|
5.8 |
|
| |
3.00 |
|
|
|
311,150 |
|
|
|
6.0 |
|
|
|
77,787 |
|
|
|
3.00 |
|
|
|
6.0 |
|
| |
3.89 |
|
|
|
99,891 |
|
|
|
0.9 |
|
|
|
99,891 |
|
|
|
3.89 |
|
|
|
0.9 |
|
| |
4.11 |
|
|
|
153,264 |
|
|
|
2.8 |
|
|
|
153,264 |
|
|
|
4.11 |
|
|
|
2.8 |
|
| |
6.66 |
|
|
|
28,608 |
|
|
|
8.2 |
|
|
|
28,608 |
|
|
|
6.66 |
|
|
|
8.2 |
|
| |
7.25 |
|
|
|
33,040 |
|
|
|
6.4 |
|
|
|
16,520 |
|
|
|
7.25 |
|
|
|
6.4 |
|
| |
18.94 |
|
|
|
12,169 |
|
|
|
4.5 |
|
|
|
11,080 |
|
|
|
18.94 |
|
|
|
4.5 |
|
| |
22.28 |
|
|
|
89,100 |
|
|
|
10 |
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
68.25 |
|
|
|
555 |
|
|
|
1.8 |
|
|
|
555 |
|
|
|
68.25 |
|
|
|
1.8 |
|
| |
108.00 |
|
|
|
7,094 |
|
|
|
0.1 |
|
|
|
7,094 |
|
|
|
108.00 |
|
|
|
0.1 |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
802,871 |
|
|
|
5.19 |
|
|
|
462,799 |
|
|
|
3.44 |
|
|
|
3.82 |
|
As at December 31, 2009 and December 31, 2008 the total unrecognized compensation expense related
to stock options granted is $1,910,000 and $740,000 respectively, which is expected to be
recognized into expense over a period of approximately 4 years.
The estimated grant date fair value of stock options vested during the years ended December 31,
2009, 2008, and 2007 was $399,000, $160,000 and $189,000 respectively.
The aggregate intrinsic value of options exercised was calculated as the difference between the
exercise price of the stock options and the fair value of the underlying common stock as of the
date of exercise. The aggregate intrinsic value of options exercised for the years ended December
31, 2009 and 2008 was $697,000 and $39,000, respectively. No options were exercised in 2007. At
December 31, 2009, the aggregate intrinsic value of the outstanding options was $12,304,000 and the
aggregate intrinsic value of the exercisable options was $8,721,000.
[d] Stock Warrants
At December 31, 2009, there were warrants outstanding to purchase 183,385 shares of common stock at
exercise prices ranging from $74.70 to $79.56 per share and expiration dates ranging from August
2010 to October 2010.
[e] Sonus Employee Stock Purchase Plan
Prior to January 2009, the Company had an employee stock purchase plan whereby employees were
permitted to contribute up to 15% of their compensation to purchase shares of the Companys common
stock at 85% of the stocks share price at the lower of the beginning or end of each six-month
offering period. Shares purchased under the plan were 222 and 2,600 in 2008 and 2007, respectively.
The plan was terminated in January 2009 and no shares were purchased under the plan in 2009.
[f] Shareholder Rights Plan
The Company has a Shareholder Rights Plan which was adopted in July 1996 and subsequently amended
in July 2002, October 2005 and August 2006 (the Rights Plan). Under the Plan the Companys Board
of Directors declared a dividend of one Preferred Stock Purchase Right (Right) for each
outstanding common share of the Company. Subject to the Rights Plan, each Right entitles the
registered holder to purchase from the Company one one-hundredth of a share of Series A Junior
Participating Preferred Stock at a exercise price of $140, subject to adjustment. These Rights
provide the holders with the right to purchase, in the event a person or group acquires 15% or more
of the Companys common stock, additional shares of the Companys common stock having a market
value equal to two times the exercise price of the Right. Pursuant to the Rights Plan, the
one-for-eighteen reverse stock split caused a proportionate adjustment of the number of Rights
associated with each share of common stock. Currently, eighteen (18) Rights are associated with
each share of common stock.
93
[g] 401(k) Plan
The Company maintains a 401(k) plan in which it provided a specified percentage match on employee
contributions. Following the Arrangement, the Board of Directors of OncoGenex amended and restated
the 401(k) plan whereas securities of the Company are no longer offered as an investment option.
This amendment prohibits the inclusion of OncoGenex shares in the 401(k) plan, as well as any match
of Company shares to employee contributions. No shares of the Company were issued subsequent to the
Arrangement, and as such no related expense was incurred.
[h] Loss per common share
Weighted average common shares outstanding for prior periods have been restated to reflect the
change in capital structure resulting from the transaction with Sonus. The following table presents
the computation of basic and diluted net loss per share:
| |
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
Years ended December 31, |
|
| (in thousands except shares and per share amounts) |
|
2009 |
|
|
2008 |
|
|
2007 |
|
| |
Numerator |
|
|
|
|
|
|
|
|
|
|
|
|
Income (loss) attributable to common shareholders as reported |
|
$ |
(5,476 |
) |
|
$ |
(6,177 |
) |
|
$ |
(11,480 |
) |
Denominator |
|
|
|
|
|
|
|
|
|
|
|
|
Weighted average number of common shares outstanding |
|
|
5,766,850 |
|
|
|
1,829,276 |
|
|
|
118,801 |
|
Basic and diluted income (loss) per common share |
|
$ |
(.95 |
) |
|
$ |
(3.38 |
) |
|
$ |
(96.63 |
) |
Earnings per share associated with $4,428 extraordinary gain |
|
$ |
|
|
|
|
2.42 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Basic and diluted income (loss) per common share excluding
extraordinary gain |
|
$ |
(.95 |
) |
|
$ |
(5.80 |
) |
|
$ |
(96.63 |
) |
As of December 31, 2009, 2008 and 2007 a total of 986,256, 906,528 and 324,231 options and
warrants, respectively, have not been included in the calculation of potential common shares as
their effect on diluted per share amounts would have been anti-dilutive.
12. RELATED PARTY TRANSACTIONS
Upon incorporation of OncoGenex Technologies, a former director assigned certain intellectual
property to the Company in exchange for 178,564 common shares (820,000 OncoGenex Technologies
shares). These common shares were recorded at a nominal amount representing the directors original
cost of the intellectual property.
The Company incurred consulting fees to a director of $163,000 and $123,000 for the years ended
December 31, 2008 and 2007. There were no related party transactions during the period ended
December 31, 2009, and no amounts were included in accounts payable and accrued liabilities as at
December 31, 2009 and 2008. All transactions were recorded at their exchange amounts.
13. COMMITMENTS AND CONTINGENCIES
Teva Pharmaceutical Industries Ltd.
In December 2009, OncoGenex Pharmaceuticals, Inc., through its wholly-owned subsidiary, OncoGenex
Technologies, entered into a Collaboration Agreement with Teva for the development and global
commercialization of OGX-011 (and related compounds). Under the Collaboration Agreement, Teva made
upfront payments in the aggregate amount of $50 million, will pay up to $370 million upon the
achievement of developmental and commercial milestones and will pay royalties at percentage rates
ranging from the mid-teens to mid-twenties on net sales. The Company is required to contribute $30
million in direct and indirect costs towards the Clinical Development Plan. $3.5 million of these
costs were incurred by OncoGenex during 2009, resulting in a remaining funding responsibility of
$26.5 million which has been recorded as Deferred Collaboration Revenue. Teva will fund all other
expenses under the Clinical Development Plan.
94
Pursuant to the Collaboration Agreement, OncoGenex and Teva agreed to collaborate in the
development and global commercialization of OGX-011. Teva received the exclusive worldwide right
and license to develop and commercialize products containing OGX-011 and related compounds (the
Licensed Products). OncoGenex has an option to co-promote OGX-011 in the United States and
Canada.
In addition to the development costs noted above, Teva is also responsible for all costs relating
to product commercialization including costs incurred in relation to the Companys co-promotion
option, except for start-up costs in advance of commercialization.
Isis Pharmaceuticals Inc. and University of British Columbia
Pursuant to license agreements the Company has with the University of British Columbia (UBC) and
Isis Pharmaceuticals Inc., the Company is obligated to pay royalties on future product sales and
milestone payments of up to $10.1 million upon the achievement of specified product development
milestones related to OGX-427 and OGX-225.
In addition, we are required to
pay to Isis 30% of all Non-Royalty Revenue we receive. Isis has disclosed in its SEC filings
that it is entitled to receive 30% of the up to $370 million in milestone payments we may receive
from Teva as part of the Collaboration Agreement; however, we believe that certain of the milestone
payments related to sales targets may qualify as Royalty Revenue, and therefore be subject to
the lesser payment obligations. No assurance can be provided that we will be entitled to receive
these milestone payments or, if we are, that the applicable amount payable to Isis will be less than
30%. We are also obligated to pay to UBC certain patent costs
and annual license maintenance fees for the extent of the patent life of CAD $8,000 per year. We
anticipate paying Isis $750,000 in 2010 upon the initiation of a phase 2 clinical trial of OGX-427
in patients with CRPC. We do not anticipate making any royalty payments to Isis in 2010.
The UBC agreements have effective dates ranging from November 1, 2001 to April 5, 2005 and each
agreement expires upon the later of 20 years from its effective date or the expiry of the last
patent licensed thereunder, unless otherwise terminated.
Unless otherwise terminated, the Isis agreements for OGX-011 and OGX-427 will continue for each
product until the later of 10 years after the date of the first commercial product sale, or the
expiration of the last to expire of any patents required to be licensed in order to use or sell the
product, unless OncoGenex Technologies abandons either OGX-011 or OGX-427 and Isis does not elect
to unilaterally continue development. The Isis agreement for OGX-225 will continue into perpetuity
unless OncoGenex Technologies abandons the product and Isis does not elect to unilaterally continue
development.
To facilitate the execution and performance of the Collaboration Agreement with Teva, OncoGenex and
Isis agreed to amend the Isis License Agreement and the Company and UBC agreed to amend the UBC
License Agreement, in each case, effective December 19 and December 20, 2009, respectively.
The amendment to the Isis License Agreement provides, among other things, that if the Company is
the subject of a change of control with a third party, where the surviving company immediately
following such change of control has the right to develop and sell the product, then (i) a
milestone payment of $20 million will be due and payable to Isis 21 days following the first
commercial sale of the product in the United States; and (ii) unless such surviving entity had
previously sublicensed the product and a royalty rate payable to Isis by the Company has been
established, the applicable royalty rate payable to Isis will thereafter be the maximum amount
payable under the Isis License Agreement. Any non-royalty milestone amounts previously paid will be
credited toward the $20 million milestone if not already paid. As a result of the $10 million
milestone payment payable to Isis in relation to the Collaboration Agreement, the remaining amount
owing in the event of change of control discussed above is a maximum of $10 million. As the Company
has now licensed the product to Teva and established a royalty rate payable to Isis, no royalty
rate adjustments would apply if Teva acquires the Company and is the surviving company. If the $30
million in advanced reimbursement of development activities has not been spent by OncoGenex prior
to the third anniversary of the Collaboration Agreement between OncoGenex and Teva, OncoGenex will
pay Isis an amount equal to 30% of any un-spent portion less $3.5 million.
95
Bayer HealthCare LLC
On August 7, 2008, Sonus completed an exclusive in-licensing agreement with Bayer HealthCare LLC
for the right to develop, commercialize or sublicense a family of compounds known as caspase
activators presently in preclinical research. Under terms of the agreement, Sonus was granted
exclusive rights to develop two core compounds for all prophylactic and therapeutic uses in humans.
Additionally, Sonus was granted rights to all other non-core compounds covered under the patents
for use in oncology.
Under the terms of the agreement, Bayer received an upfront license fee of $450,000. OncoGenex will
make annual payments to Bayer on the anniversary date (Anniversary Payments), with an initial
payment of $100,000. The payments will increase by $25,000 each year until the initiation of the
first phase 3 clinical trial, at which point the Anniversary Payments reset to $100,000 each year
and increase by $25,000 until the Company achieves either the first New Drug Application filing in
the United States or the European Union. OncoGenex is obligated to pay royalties on net future product sales in addition
to aggregate milestone payments of up to $14,000,000 for clinical
development and regulatory milestones. No milestone payments are triggered prior to the initiation
of a phase 3 clinical trial. OncoGenex has the option to terminate this contract upon 60 days
written notice to Bayer.
Lease Arrangements
The Company has an operating lease agreement for office space in Vancouver, Canada, which expires
in March 2011.
Future minimum annual lease payments under the Vancouver lease are as follows (in thousands):
| |
|
|
|
|
2010 |
|
$ |
181 |
|
2011 |
|
|
45 |
|
|
|
|
|
Total |
|
$ |
226 |
|
In November 2006, prior to the Arrangement (note 5), Sonus entered into a non-cancellable operating
lease agreement for office space in Bothell, Washington, expiring in 2017. In connection with the
new lease, Sonus was required to provide a cash security deposit of approximately $497,000, which
is included in Other Long Term Assets. In addition, the lease stipulates the Company must issue a
standby letter of credit for approximately $500,000 which is expected to be issued during 2010. The
Company is currently in the process of evaluating opportunities to exit or sublet portions of the
leased space and has recorded a liability in the excess facilities lease charge of $4,645,000 as at
December 31, 2009 (note 8).
96
If the Company is unable to exit or sublet portions of this leased space, the future minimum annual
lease payments are as follows (in thousands):
| |
|
|
|
|
2010 |
|
$ |
1,995 |
|
2011 |
|
|
2,055 |
|
2012 |
|
|
2,117 |
|
2013 |
|
|
2,180 |
|
2014 |
|
|
2,245 |
|
remainder |
|
|
7,150 |
|
|
|
|
|
Total |
|
$ |
17,742 |
|
Consolidated rent expense relating to both the Vancouver, Canada and Bothell, Washington offices
for years ended December 31, 2009, 2008, and 2007 was $2,408,000, $801,000, and $220,000
respectively.
Guarantees and Indemnifications
OncoGenex indemnifies its officers and directors for certain events or occurrences, subject to
certain limits, while the officer or director is or was serving at our request in such capacity.
The term of the indemnification period is equal to the officers or directors lifetime.
The maximum amount of potential future indemnification is unlimited; however, we have obtained
director and officer insurance that limits our exposure and may enable it to recover a portion of
any future amounts paid. We believe that the fair value of these indemnification obligations is
minimal. Accordingly, we have not recognized any liabilities relating to these obligations as of
December 31, 2009.
We have certain agreements with certain organizations with which we do business that contain
indemnification provisions pursuant to which we typically agree to indemnify the party against
certain types of third-party claims. We accrue for known indemnification issues when a loss is
probable and can be reasonably estimated. There were no accruals for or expenses related to
indemnification issues for any period presented.
14. COMPREHENSIVE INCOME (LOSS)
| |
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
Year ended December 31, |
|
| (in thousands) |
|
2009 |
|
|
2008 |
|
|
2007 |
|
| |
|
$ |
|
|
$ |
|
|
$ |
|
Loss for the period |
|
|
5,476 |
|
|
|
4,204 |
|
|
|
8,536 |
|
Unrealized gain (loss) on cash
equivalents and marketable securities |
|
|
|
|
|
|
|
|
|
|
3 |
|
Unrealized gain (loss) on foreign exchange |
|
|
|
|
|
|
(3 |
) |
|
|
557 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
5,476 |
|
|
|
4,207 |
|
|
|
7,976 |
|
97
15. QUARTERLY FINANCIAL INFORMATION (UNAUDITED)
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
Quarter Ended |
|
| |
|
Dec. 31 |
|
|
Sept. 30 |
|
|
Jun. 30 |
|
|
Mar. 31 |
|
| |
|
(in thousands, except per share amounts) |
|
2009 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Collaboration revenue |
|
$ |
25,539 |
|
|
$ |
|
|
|
$ |
|
|
|
$ |
|
|
Research and development |
|
$ |
17,365 |
|
|
$ |
1,513 |
|
|
$ |
3,588 |
|
|
$ |
1,694 |
|
General and administrative |
|
$ |
1,291 |
|
|
$ |
885 |
|
|
$ |
1,003 |
|
|
$ |
782 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total expenses |
|
$ |
18,656 |
|
|
$ |
2,398 |
|
|
$ |
4,591 |
|
|
$ |
2,476 |
|
Other income (expense) |
|
$ |
(3 |
) |
|
$ |
29 |
|
|
$ |
34 |
|
|
$ |
57 |
|
Tax expense (recovery) |
|
$ |
2,999 |
|
|
$ |
16 |
|
|
$ |
6 |
|
|
$ |
(10 |
) |
Net loss (income) attributable to common
shareholders |
|
$ |
(3,881 |
) |
|
$ |
2,385 |
|
|
$ |
4,563 |
|
|
$ |
2,409 |
|
Net loss (income) per share*: |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Basic and diluted |
|
$ |
(0.64 |
) |
|
$ |
0.40 |
|
|
$ |
0.82 |
|
|
$ |
0.43 |
|
2008 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Research and development |
|
$ |
4,198 |
|
|
$ |
1,639 |
|
|
$ |
1,108 |
|
|
$ |
874 |
|
General and administrative |
|
$ |
1,050 |
|
|
$ |
1,024 |
|
|
$ |
646 |
|
|
$ |
573 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total expenses |
|
$ |
5,248 |
|
|
$ |
2,663 |
|
|
$ |
1,754 |
|
|
$ |
1,447 |
|
Other income (expense) |
|
$ |
334 |
|
|
$ |
297 |
|
|
$ |
(213 |
) |
|
$ |
4 |
|
Tax expense (recovery) |
|
$ |
41 |
|
|
$ |
(2,515 |
) |
|
$ |
201 |
|
|
$ |
214 |
|
Redeemable preferred share accretion |
|
$ |
|
|
|
$ |
417 |
|
|
$ |
780 |
|
|
$ |
776 |
|
Extraordinary gain |
|
$ |
|
|
|
$ |
4,428 |
|
|
$ |
|
|
|
$ |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net loss (income) attributable to common
shareholders |
|
$ |
4,955 |
|
|
$ |
(4,160 |
) |
|
$ |
2,949 |
|
|
$ |
2,433 |
|
Net income (loss) per share*: |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Basic and diluted |
|
$ |
(0.99 |
) |
|
$ |
2.02 |
|
|
$ |
(24.81 |
) |
|
$ |
(20.48 |
) |
| |
|
|
| * |
|
Quarterly EPS may not add to annual figure due to rounding. |
16. SUBSEQUENT EVENTS
The Company has performed an evaluation of events occurring subsequent to year end. Based on our
evaluation, no material events have occurred requiring financial statement disclosure.
98
ITEM 9. CHANGES IN AND DISAGREEMENTS WITH ACCOUNTANTS ON ACCOUNTING AND FINANCIAL DISCLOSURE
None.
ITEM 9A. CONTROLS AND PROCEDURES
Evaluation of Disclosure Controls and Procedures
The Company maintains disclosure controls and procedures that are designed to ensure that material
information required to be disclosed in the Companys periodic reports filed or submitted under the
Exchange Act, is recorded, processed, summarized and reported within the time periods specified in
the SECs rules and forms. The Companys disclosure controls and procedures are also designed to
ensure that information required to be disclosed in the reports the Company files or submits under
the Exchange Act is accumulated and communicated to the Companys management, including its
principal executive officer and principal financial officer as appropriate, to allow timely
decisions regarding required disclosure.
The Company carried out an evaluation, under the supervision and with the participation of the
Companys management, including the principal executive officer and the principal financial
officer, of the effectiveness of the design and operation of the disclosure controls and
procedures, as defined in Rules 13a-15(e) and 15d-15(e) under the Exchange Act. Based upon that
evaluation, the Companys principal executive officer and principal financial officer concluded
that the Companys disclosure controls and procedures were effective, as of the end of the period
covered by this report.
Changes in Internal Control Over Financial Reporting
The Company has not made any changes to our internal control over financial reporting (as defined
in Rule 13a-15(f) and 15d-15(f) under the Exchange Act) during the quarter ended December 31, 2009
that have materially affected, or are reasonably likely to materially affect, the Companys
internal control over financial reporting.
Managements Report on Internal Control over Financial Reporting
The management of the Company is responsible for establishing and maintaining adequate internal
control over financial reporting, as defined in Rule 13a-15(f) under the Exchange Act. The
Companys internal control over financial reporting is a process designed under the supervision of
the Companys principal executive and principal financial officers to provide reasonable assurance
regarding the reliability of financial reporting and the preparation of the Companys financial
statements for external reporting purposes in accordance with U.S. generally accepted accounting
principles.
As of December 31, 2009, management assessed the effectiveness of the Companys internal control
over financial reporting based on the framework established in Internal ControlIntegrated
Framework issued by the Committee of Sponsoring Organizations of the Treadway Commission (COSO).
Based on this evaluation, management has determined that the Companys internal control over
financial reporting was effective as of December 31, 2009.
Because of its inherent limitations, internal control over financial reporting may not prevent or
detect misstatements. Also, projections of any evaluation of effectiveness to future periods are
subject to the risk that controls may become inadequate because of changes in conditions, or that
the degree of compliance with the policies or procedures may deteriorate.
The effectiveness of our internal control over financial reporting as of December 31, 2009 has been
audited by Ernst & Young LLP, an independent registered public accounting firm, as stated in their
report which is included above.
ITEM 9B. OTHER INFORMATION
Not applicable.
99
PART III
ITEM 10. DIRECTORS, EXECUTIVE OFFICERS AND CORPORATE GOVERNANCE
The information required hereunder is incorporated by reference from our definitive Proxy Statement
to be filed within 120 days of December 31, 2009 and delivered to shareholders in connection with
our 2010 Annual Meeting of Shareholders.
ITEM 11. EXECUTIVE COMPENSATION
The information required hereunder is incorporated by reference from our definitive Proxy Statement
to be filed within 120 days of December 31, 2009 and delivered to shareholders in connection with
our 2010 Annual Meeting of Shareholders.
ITEM 12. SECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT AND RELATED STOCKHOLDER
MATTERS
The following table sets forth certain information regarding our equity compensation plans as of
December 31, 2009:
| |
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
(a) |
|
|
(b) |
|
|
(c) |
|
| |
|
|
|
|
|
|
|
|
|
Number of securities |
|
| |
|
|
|
|
|
|
|
|
|
remaining |
|
| |
|
|
|
|
|
|
|
|
|
available for future issuance |
|
| |
|
Number of securities to be |
|
|
Weighted-average |
|
|
under equity compensation |
|
| |
|
issued upon exercise of |
|
|
exercise price of |
|
|
plans |
|
| |
|
outstanding |
|
|
outstanding |
|
|
(excluding securities |
|
| |
|
options, warrants |
|
|
options, warrants |
|
|
reflected in |
|
| Plan category |
|
and rights |
|
|
and rights |
|
|
column (a) |
|
Equity compensation plans approved by security holders |
|
|
781,452 |
(1) |
|
$ |
7.98 |
(1) |
|
|
53,684 |
(1) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Equity compensation plans not approved by security
holders(2)
|
|
|
21,419 |
|
|
|
2.69 |
|
|
|
0 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total |
|
|
802,871 |
|
|
$ |
6.95 |
|
|
|
53,684 |
|
| |
|
|
| (1) |
|
As at December 31, 2009, the Company maintained the following equity compensation
plans, which were adopted by Sonus prior to the Arrangement: (a) the Incentive Stock
Option, Nonqualified Stock Option and Restricted Stock Purchase Plan 1991, (b) the 1999
Nonqualified Stock Incentive Plan (the 1999 Plan), (c) the 2000 Stock Incentive Plan, (d)
the 2007 Performance Incentive Plan, and (e) the OncoGenex Technologies Amended and
Restated Stock Option Plan. |
| |
| (2) |
|
The 1999 Plan is a broad-based plan for which shareholder approval was not required or
obtained. On February 11, 2009, the 1999 Plan terminated in accordance with its terms. All
stock options outstanding as of such time will continue in effect in accordance with their
respective terms. Stock options granted under the 1999 Plan were generally granted with an
exercise price equal to fair market value on the date of grant. |
The remaining information required hereunder is incorporated by reference from our definitive Proxy
Statement to be filed within 120 days of December 31, 2009 and delivered to shareholders in
connection with its 2010 Annual Meeting of Shareholders.
100
ITEM 13. CERTAIN RELATIONSHIPS AND RELATED TRANSACTIONS, AND DIRECTOR INDEPENDENCE
The information required hereunder is incorporated by reference from our definitive Proxy Statement
to be filed within 120 days of December 31, 2009 and delivered to shareholders in connection with
its 2010 Annual Meeting of Shareholders.
ITEM 14. PRINCIPAL ACCOUNTANT FEES AND SERVICES
The information required hereunder is incorporated by reference from our definitive Proxy Statement
to be filed within 120 days of December 31, 2009 and delivered to shareholders in connection with
its 2010 Annual Meeting of Shareholders.
PART IV
ITEM 15. EXHIBITS AND FINANCIAL STATEMENT SCHEDULES
(a)
(1) Financial Statements
| |
|
|
|
|
Report of Independent Registered Public Accounting Firm |
|
|
66 |
|
|
|
|
|
|
Consolidated Balance Sheets as of December 31, 2009 and 2008 |
|
|
68 |
|
|
|
|
|
|
Consolidated Statements of Loss for the years ended December 31, 2009,
2008, and 2007 |
|
|
69 |
|
|
|
|
|
|
Consolidated Statements of Shareholders Equity for the years ended
December 31, 2009, 2008, and 2007 |
|
|
70 |
|
|
|
|
|
|
Consolidated Statements of Cash Flows for the years ended December 31,
2009, 2008, and 2007 |
|
|
71 |
|
|
|
|
|
|
Consolidated Notes to the Financial Statements |
|
|
72 |
|
(2)
All schedules are omitted because they are not required or the required information is included in
the financial statements or notes thereto.
101
(3)
Exhibits
| |
|
|
|
|
| Exhibit |
|
|
| Number |
|
Description |
| |
|
|
|
|
| |
2.1 |
(1) |
|
Arrangement Agreement between the Company and OncoGenex Technologies Inc. dated May 27, 2008 |
| |
|
|
|
|
| |
2.2 |
(2) |
|
First Amendment to Arrangement Agreement between the Company and OncoGenex Technologies Inc. dated August 11, 2008 |
| |
|
|
|
|
| |
2.3 |
(2) |
|
Second Amendment to Arrangement Agreement between the Company and OncoGenex Technologies Inc. dated August 15, 2008 |
| |
|
|
|
|
| |
3.1 |
(3) |
|
Amended and Restated Certificate of Incorporation (As Amended Through October 17, 1995) |
| |
|
|
|
|
| |
3.2 |
(4) |
|
Certificate of Amendment to Certificate of Incorporation filed on May 6, 1999 |
| |
|
|
|
|
| |
3.3 |
(5) |
|
Certificate of Correction filed on March 9, 2009 to Certificate of Amendment filed on May 6, 1999 |
| |
|
|
|
|
| |
3.4 |
(6) |
|
Certificate of Amendment to Certificate of Incorporation filed on May 7, 2004 |
| |
|
|
|
|
| |
3.5 |
(5) |
|
Certificate of Correction filed on March 9, 2009 to Certificate of Amendment filed on May 7, 2004 |
| |
|
|
|
|
| |
3.6 |
(2) |
|
Certificate of Amendment to Certificate of Incorporation of Sonus Pharmaceuticals Inc., effective August 20, 2008 |
| |
|
|
|
|
| |
3.7 |
(7) |
|
Third Amended and Restated Bylaws of Oncogenex Pharmaceuticals, Inc. |
| |
|
|
|
|
| |
4.1 |
(2) |
|
Specimen Certificate of Common Stock |
| |
|
|
|
|
| |
4.2 |
(8) |
|
Amended and Restated Rights Agreement dated as of July 24, 2002 between Sonus Pharmaceuticals Inc. and U.S. Stock
Transfer Corporation |
| |
|
|
|
|
| |
4.3 |
(9) |
|
First Amendment to Amended and Restated Rights Agreement dated as of October 17, 2005 between Sonus
Pharmaceuticals Inc. and U.S. Stock Transfer Corporation |
| |
|
|
|
|
| |
4.4 |
(10) |
|
Second Amendment to Amended and Restated Rights Agreement dated as of August 10, 2006 between Sonus
Pharmaceuticals Inc. and U.S. Stock Transfer Corporation |
| |
|
|
|
|
| |
4.5 |
(11) |
|
Third Amendment to Amended and Restated Rights Agreement dated May 27, 2008 between Sonus Pharmaceuticals Inc. and
Computershare Trust Company, N.A. |
| |
|
|
|
|
| |
4.6 |
(1) |
|
Form of Escrow Agreement between the Company, Computershare Trust Company of Canada and former shareholders and
debentureholders of OncoGenex Technologies Inc. |
| |
|
|
|
|
| |
4.7 |
(1) |
|
Form of OncoGenex Voting Agreement |
| |
|
|
|
|
| |
4.8 |
(1) |
|
Form of Sonus Voting Agreement |
| |
|
|
|
|
| |
10.1 |
(3) |
|
Sonus Pharmaceuticals, Inc. Incentive Stock Option, Nonqualified Stock Option and Restricted Stock Purchase Plan
1991 (the 1991 Plan), as amended |
102
| |
|
|
|
|
| Exhibit |
|
|
| Number |
|
Description |
| |
|
|
|
|
| |
10.2 |
(3) |
|
Form of Incentive Option Agreement (pertaining to the 1991 Plan) |
| |
|
|
|
|
| |
10.3 |
(3) |
|
Form of Sonus Pharmaceuticals, Inc. Nonqualified Stock Option Agreement under the 1991 Plan |
| |
|
|
|
|
| |
10.4 |
(4) |
|
Sonus Pharmaceuticals, Inc. 1999 Nonqualified Stock Incentive Plan (the 1999 Plan) |
| |
|
|
|
|
| |
10.5 |
(4) |
|
Form of Sonus Pharmaceuticals, Inc. Nonqualified Stock Option Agreement under the 1999 Plan |
| |
|
|
|
|
| |
10.6 |
(4) |
|
Form of Sonus Pharmaceuticals, Inc. Restricted Stock Purchase Agreement under the 1999 Plan |
| |
|
|
|
|
| |
10.7 |
(12) |
|
Sonus Pharmaceuticals, Inc. 2000 Stock Incentive Plan (the 2000 Plan) |
| |
|
|
|
|
| |
10.8 |
(13) |
|
First Amendment to Sonus Pharmaceuticals, Inc. 2000 Plan |
| |
|
|
|
|
| |
10.9 |
(12) |
|
Form of Sonus Pharmaceuticals, Inc. Stock Option Agreement (pertaining to the 2000 Plan) |
| |
|
|
|
|
| |
10.10 |
(14) |
|
Sonus Pharmaceuticals, Inc. 2007 Performance Incentive Plan (the 2007 Plan) |
| |
|
|
|
|
| |
10.11 |
(15) |
|
Form of Sonus Pharmaceuticals, Inc. Stock Option Agreement (pertaining to the 2007 Plan) |
| |
|
|
|
|
| |
10.12 |
(15) |
|
Form of Sonus Pharmaceuticals, Inc. Restricted Stock Purchase Agreement under the 2007 Plan |
| |
|
|
|
|
| |
10.13 |
(16) |
|
OncoGenex Technologies Inc. Amended and Restated Stock Option Plan |
| |
|
|
|
|
| |
10.14 |
(17) |
|
Stock Option Assumption, Amending and Confirmation Agreement dated as of August 21, 2008 between the Company and
OncoGenex Technologies Inc. |
| |
|
|
|
|
| |
10.15 |
(18) |
|
OncoGenex Pharmaceuticals, Inc. Short Term Incentive Awards Program |
| |
|
|
|
|
| |
10.16 |
(18) |
|
Agreement and Consent Form (related to the Short Term Incentive Awards Program) |
| |
|
|
|
|
| |
10.17 |
(3) |
|
Form of Indemnification Agreement for Officers and Directors of the Company |
| |
|
|
|
|
| |
10.18 |
(16) |
|
Form of Indemnification Agreement between OncoGenex Technologies Inc. and each of Scott Cormack, Stephen Anderson
and Cindy Jacobs |
| |
|
|
|
|
| |
10.19 |
(16) |
|
Form of Indemnification Agreement between OncoGenex Technologies Inc. and Neil Clendeninn |
| |
|
|
|
|
| |
10.20 |
(2) |
|
Executive Termination Agreement and General Release dated August 21, 2008 between the Company and Michael Martino |
| |
|
|
|
|
| |
10.21 |
(2) |
|
Executive Termination Agreement and General Release dated August 21, 2008 between the Company and Alan Fuhrman |
103
| |
|
|
|
|
| Exhibit |
|
|
| Number |
|
Description |
| |
|
|
|
|
| |
10.22 |
(19) |
|
Employment Agreement between OncoGenex Technologies Inc. and the Company and Scott Cormack dated as of November
4, 2009 |
| |
|
|
|
|
| |
10.23 |
(19) |
|
Employment Agreement between OncoGenex Technologies Inc. and the Company and Stephen Anderson dated as of
November 4, 2009 |
| |
|
|
|
|
| |
10.24 |
(20) |
|
Amendment dated February 24, 2010 to the Employment Agreement between OncoGenex Technologies Inc. and Stephen
Anderson |
| |
|
|
|
|
| |
10.25 |
(19) |
|
Employment Agreement between the Company and Cindy Jacobs dated as of November 3, 2009 |
| |
|
|
|
|
| |
10.26 |
(21) |
|
Employment Agreement dated October 14, 2008 between OncoGenex Technologies Inc. and Cameron Lawrence |
| |
|
|
|
|
| |
10.27 |
(21) |
|
Employment Amending Agreement dated January 1, 2009 between OncoGenex Technologies Inc. and Cameron Lawrence |
| |
|
|
|
|
| |
10.28 |
(22) |
|
Securities Purchase Agreement dated as of August 15, 2005 by and among Sonus Pharmaceuticals Inc. and the
investors named therein |
| |
|
|
|
|
| |
10.29 |
(22) |
|
Form of Purchase Warrant related to the Securities Purchase Agreement |
| |
|
|
|
|
| |
10.30 |
(23) |
|
Form of Purchase Warrant issued to Schering AG |
| |
|
|
|
|
| |
10.31 |
(22) |
|
Registration Rights Agreement dated as of August 15, 2005 by and among Sonus Pharmaceuticals Inc. and the
investors named therein |
| |
|
|
|
|
| |
10.32 |
(24) |
|
Lease by and between BMR-217th Place LLC and the Company dated as of November 21, 2006 |
| |
|
|
|
|
| |
10.33 |
(25) |
|
First Amendment to Lease by and between BMR-217th Place LLC and the Company dated as of August 17, 2007 |
| |
|
|
|
|
| |
10.34 |
(26) |
|
Second Amendment to Lease by and between BMR-217th Place LLC and the Company dated as
of January 28, 2008 |
| |
|
|
|
|
| |
10.35 |
(6) |
|
Amended and Restated License Agreement effective as of July 2, 2008 by and between
OncoGenex Technologies Inc. and Isis Pharmaceuticals, Inc. (OGX-011)* |
| |
|
|
|
|
| |
10.36 |
|
|
Letter Agreement Regarding Certain Sublicense Consideration for OGX-011 between OncoGenex Technologies Inc. and
Isis Pharmaceuticals, Inc. dated December 18, 2009 |
| |
|
|
|
|
| |
10.37 |
|
|
Amendment No. 1 to Amended and Restated License Agreement between OncoGenex Technologies Inc. and Isis
Pharmaceuticals, Inc. dated December 19, 2009 (OGX-011)* |
| |
|
|
|
|
| |
10.38 |
(27) |
|
License Agreement between OncoGenex Technologies Inc. and the University of British
Columbia effective as of November 1, 2001, and Amending Agreement dated as of August 30, 2006 (OGX-011)* |
| |
|
|
|
|
| |
10.39 |
(2) |
|
Second Amending Agreement and Consent as of August 7, 2008 between the University of British Columbia and
OncoGenex Technologies Inc. (OGX-011) |
| |
|
|
|
|
| |
10.40 |
|
|
Third Amending Agreement to the License Agreement between OncoGenex Technologies Inc and the University of
British Columbia dated as of December 20, 2009 (OGX-011)* |
104
| |
|
|
|
|
| Exhibit |
|
|
| Number |
|
Description |
| |
|
|
|
|
| |
10.41 |
(27) |
|
Collaboration and License Agreement between OncoGenex Technologies Inc. and Isis
Pharmaceuticals, Inc. effective as of January 5, 2005 (OGX-427)* |
| |
|
|
|
|
| |
10.42 |
(27) |
|
License Agreement between OncoGenex Technologies Inc. and the University of British
Columbia effective as of April 5, 2005, and Amending Agreement dated as of August 30, 2006 (OGX-427)* |
| |
|
|
|
|
| |
10.43 |
(2) |
|
Second Amending Agreement as of August 7, 2008 between the University of British Columbia and OncoGenex
Technologies Inc. (OGX-427) |
| |
|
|
|
|
| |
10.44 |
|
|
Collaboration and License Agreement between OncoGenex Technologies Inc. and Teva Pharmaceutical Industries Ltd.
dated as of December 20, 2009 (OGX-011)* |
| |
|
|
|
|
| |
21.1 |
|
|
Subsidiaries of the Registrant |
| |
|
|
|
|
| |
23.1 |
|
|
Consent of Ernst & Young LLP |
| |
|
|
|
|
| |
31.1 |
|
|
Certification of President and Chief Executive Officer pursuant to Rule 13a-14(a) or 15d-14(a) of the Securities
Exchange Act of 1934, as adopted pursuant to Section 302 of the Sarbanes-Oxley Act of 2002 |
| |
|
|
|
|
| |
31.2 |
|
|
Certification of Chief Financial Officer pursuant to Rule 13a-14(a) or 15d-14(a) of the Securities Exchange Act
of 1934, as adopted pursuant to Section 302 of the Sarbanes-Oxley Act of 2002 |
| |
|
|
|
|
| |
32.1 |
|
|
Certification of President and Chief Executive Officer pursuant to 18 U.S.C. Section 1350, as adopted pursuant to
Section 906 of the Sarbanes-Oxley Act of 2002 |
| |
|
|
|
|
| |
32.2 |
|
|
Certification of Chief Financial Officer pursuant to 18 U.S.C. Section 1350, as adopted pursuant to Section 906
of the Sarbanes-Oxley Act of 2002 |
| |
|
|
| |
|
Schedules and similar attachments to the Arrangement Agreement have been omitted pursuant to
Item 601(b)(2) of Regulation S-K. Registrant will furnish supplementally a copy of any omitted schedule or similar attachment to the SEC upon request. |
| |
| * |
|
Confidential portions of this exhibit have been omitted and filed separately with the Commission pursuant to an application for Confidential Treatment under Rule 24b-2 promulgated under the Securities
Exchange Act of 1934, as amended. |
| |
| (1) |
|
Incorporated by reference to the Companys proxy statement on Schedule 14A filed on July 3, 2008. |
| |
| (2) |
|
Incorporated by reference to the Companys quarterly report on Form 10-Q for the quarter ended
September 30, 2008. |
| |
| (3) |
|
Incorporated by reference to the Companys Registration Statement on Form S-1, Reg. No. 33-96112. |
| |
| (4) |
|
Incorporated by reference to Companys quarterly report on Form 10-Q for the quarter ended March 31, 1999. |
| |
| (5) |
|
Incorporated by reference to the Companys current report on Form 8-K filed on March 11, 2009. |
| |
| (6) |
|
Incorporated by reference to the Companys annual report on Form 10-K for the year ended December 31, 2008. |
105
| |
|
|
| (7) |
|
Incorporated by reference to the Companys current report on Form 8-K filed on October 30, 2008. |
| |
| (8) |
|
Incorporated by reference to the Companys amended Form 8-A filed on July 25, 2002. |
| |
| (9) |
|
Incorporated by reference to the Companys amended Form 8-A filed on October 18, 2005. |
| |
| (10) |
|
Incorporated by reference to the Companys amended Form 8-A filed on August 14, 2006. |
| |
| (11) |
|
Incorporated by reference to the Companys current report on Form 8-K filed on May 30, 2008. |
| |
| (12) |
|
Incorporated by reference to the Companys quarterly report on Form 10-Q for the quarter ended
June 30, 2000. |
| |
| (13) |
|
Incorporated by reference to the Companys quarterly report on Form 10-Q for the quarter ended
September 30, 2006. |
| |
| (14) |
|
Incorporated by reference to the Companys proxy statement on Schedule 14A filed on April 3, 2007. |
| |
| (15) |
|
Incorporated by reference to the Companys quarterly report on Form 10-Q for the quarter ended
September 30, 2007. |
| |
| (16) |
|
Incorporated by reference to the OncoGenex Technologies Inc. registration statement on Form F-1 filed on December 13, 2006. |
| |
| (17) |
|
Incorporated by reference to the Companys registration statement on Form S-8 filed on August 26, 2008. |
| |
| (18) |
|
Incorporated by reference to the Companys current report on Form 8-K filed on April 2, 2009. |
| |
| (19) |
|
Incorporated by reference to the Companys quarterly report on Form 10-Q for the quarter ended September 30, 2009. |
| |
| (20) |
|
Incorporated by reference to the Companys current report on Form 8-K filed February 25, 2010. |
| |
| (21) |
|
Incorporated by reference to the Companys current report on Form 8-K filed March 1, 2010. |
| |
| (22) |
|
Incorporated by reference to the Companys current report on Form 8-K filed on August 18, 2005. |
| |
| (23) |
|
Incorporated by reference to the Schedule 13D filed by Schering Berlin Venture Corporation on October 31, 2005. |
| |
| (24) |
|
Incorporated by reference to the Companys annual report on Form 10-K for the year ended December 31, 2006. |
| |
| (25) |
|
Incorporated by reference to the Companys annual report on Form 10-K for the year ended December 31, 2007. |
| |
| (26) |
|
Incorporated by reference to the Companys quarterly report on Form 10-Q for the quarter ended March 31, 2008. |
| |
| (27) |
|
Incorporated by reference to the OncoGenex Technologies Inc. registration statement on Form F-1,
Amendment No. 1, filed on January 29, 2007. |
106
SIGNATURES
Pursuant to the requirements of Section 13 or 15(d) of the Securities Exchange Act of 1934,
the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto
duly authorized.
| |
|
|
|
|
| |
ONCOGENEX PHARMACEUTICALS, INC.
(Registrant)
|
|
| Date: March 8, 2010 |
By: |
/s/ SCOTT CORMACK
|
|
| |
|
Scott Cormack |
|
| |
|
Chief Executive Officer and President
(principal executive officer) |
|
Pursuant to the requirements of the Securities Exchange Act of 1934, this Report has been
signed below by the following persons on behalf of the registrant and in the capacities and on the
dates indicated.
| |
|
|
|
|
|
|
|
|
By:
|
|
/s/ SCOTT CORMACK
|
|
|
|
Chief Executive Officer, President and
|
|
Date: March 8, 2010 |
|
|
Scott Cormack |
|
|
|
Director (principal executive officer) |
|
|
|
|
|
|
|
|
|
|
|
By:
|
|
/s/ CAMERON LAWRENCE
|
|
|
|
Principal financial officer and
|
|
Date: March 8, 2010 |
|
|
Cameron Lawrence |
|
|
|
principal accounting officer |
|
|
|
|
|
|
|
|
|
|
|
By:
|
|
/s/ MICHAEL MARTINO
|
|
|
|
Director
|
|
Date: March 8, 2010 |
|
|
Michael Martino |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
By:
|
|
/s/ MICHELLE BURRIS
|
|
|
|
Director
|
|
Date: March 8, 2010 |
|
|
Michelle Burris |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
By:
|
|
/s/ DWIGHT WINSTEAD
|
|
|
|
Director
|
|
Date: March 8, 2010 |
|
|
Dwight Winstead |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
By:
|
|
/s/ NEIL CLENDENINN
|
|
|
|
Director
|
|
Date: March 8, 2010 |
|
|
Neil Clendeninn |
|
|
|
|
|
|
107
(3)
Exhibits
| |
|
|
|
|
| Exhibit |
|
|
| Number |
|
Description |
| |
|
|
|
|
| |
2.1 |
(1) |
|
Arrangement Agreement between the Company and OncoGenex Technologies Inc. dated
May 27, 2008 |
| |
|
|
|
|
| |
2.2 |
(2) |
|
First Amendment to Arrangement Agreement between the Company and OncoGenex
Technologies Inc. dated August 11, 2008 |
| |
|
|
|
|
| |
2.3 |
(2) |
|
Second Amendment to Arrangement Agreement between the Company and OncoGenex
Technologies Inc. dated August 15, 2008 |
| |
|
|
|
|
| |
3.1 |
(3) |
|
Amended and Restated Certificate of Incorporation (As Amended Through October 17, 1995) |
| |
|
|
|
|
| |
3.2 |
(4) |
|
Certificate of Amendment to Certificate of Incorporation filed on May 6, 1999 |
| |
|
|
|
|
| |
3.3 |
(5) |
|
Certificate of Correction filed on March 9, 2009 to Certificate of Amendment filed on
May 6, 1999 |
| |
|
|
|
|
| |
3.4 |
(6) |
|
Certificate of Amendment to Certificate of Incorporation filed on May 7, 2004 |
| |
|
|
|
|
| |
3.5 |
(5) |
|
Certificate of Correction filed on March 9, 2009 to Certificate of Amendment filed on
May 7, 2004 |
| |
|
|
|
|
| |
3.6 |
(2) |
|
Certificate of Amendment to Certificate of Incorporation of Sonus Pharmaceuticals
Inc., effective August 20, 2008 |
| |
|
|
|
|
| |
3.7 |
(7) |
|
Third Amended and Restated Bylaws of Oncogenex Pharmaceuticals, Inc. |
| |
|
|
|
|
| |
4.1 |
(2) |
|
Specimen Certificate of Common Stock |
| |
|
|
|
|
| |
4.2 |
(8) |
|
Amended and Restated Rights Agreement dated as of July 24, 2002 between Sonus
Pharmaceuticals Inc. and U.S. Stock Transfer Corporation |
| |
|
|
|
|
| |
4.3 |
(9) |
|
First Amendment to Amended and Restated Rights Agreement dated as of October 17, 2005
between Sonus Pharmaceuticals Inc. and U.S. Stock Transfer Corporation |
| |
|
|
|
|
| |
4.4 |
(10) |
|
Second Amendment to Amended and Restated Rights Agreement dated as of August 10, 2006
between Sonus Pharmaceuticals Inc. and U.S. Stock Transfer Corporation |
| |
|
|
|
|
| |
4.5 |
(11) |
|
Third Amendment to Amended and Restated Rights Agreement dated May 27, 2008 between
Sonus Pharmaceuticals Inc. and Computershare Trust Company, N.A. |
| |
|
|
|
|
| |
4.6 |
(1) |
|
Form of Escrow Agreement between the Company, Computershare Trust Company of Canada
and former shareholders and debentureholders of OncoGenex Technologies Inc. |
| |
|
|
|
|
| |
4.7 |
(1) |
|
Form of OncoGenex Voting Agreement |
| |
|
|
|
|
| |
4.8 |
(1) |
|
Form of Sonus Voting Agreement |
| |
|
|
|
|
| |
10.1 |
(3) |
|
Sonus Pharmaceuticals, Inc. Incentive Stock Option, Nonqualified Stock Option and
Restricted Stock Purchase Plan 1991 (the 1991 Plan), as amended |
| |
|
|
|
|
| Exhibit |
|
|
| Number |
|
Description |
| |
|
|
|
|
| |
10.2 |
(3) |
|
Form of Incentive Option Agreement (pertaining to the 1991 Plan) |
| |
|
|
|
|
| |
10.3 |
(3) |
|
Form of Sonus Pharmaceuticals, Inc. Nonqualified Stock Option Agreement under the
1991 Plan |
| |
|
|
|
|
| |
10.4 |
(4) |
|
Sonus Pharmaceuticals, Inc. 1999 Nonqualified Stock Incentive Plan (the 1999 Plan) |
| |
|
|
|
|
| |
10.5 |
(4) |
|
Form of Sonus Pharmaceuticals, Inc. Nonqualified Stock Option Agreement under the
1999 Plan |
| |
|
|
|
|
| |
10.6 |
(4) |
|
Form of Sonus Pharmaceuticals, Inc. Restricted Stock Purchase Agreement under the
1999 Plan |
| |
|
|
|
|
| |
10.7 |
(12) |
|
Sonus Pharmaceuticals, Inc. 2000 Stock Incentive Plan (the 2000 Plan) |
| |
|
|
|
|
| |
10.8 |
(13) |
|
First Amendment to Sonus Pharmaceuticals, Inc. 2000 Plan |
| |
|
|
|
|
| |
10.9 |
(12) |
|
Form of Sonus Pharmaceuticals, Inc. Stock Option Agreement (pertaining to the 2000
Plan) |
| |
|
|
|
|
| |
10.10 |
(14) |
|
Sonus Pharmaceuticals, Inc. 2007 Performance Incentive Plan (the 2007 Plan) |
| |
|
|
|
|
| |
10.11 |
(15) |
|
Form of Sonus Pharmaceuticals, Inc. Stock Option Agreement (pertaining to the 2007
Plan) |
| |
|
|
|
|
| |
10.12 |
(15) |
|
Form of Sonus Pharmaceuticals, Inc. Restricted Stock Purchase Agreement under the
2007 Plan |
| |
|
|
|
|
| |
10.13 |
(16) |
|
OncoGenex Technologies Inc. Amended and Restated Stock Option Plan |
| |
|
|
|
|
| |
10.14 |
(17) |
|
Stock Option Assumption, Amending and Confirmation Agreement dated as of August 21,
2008 between the Company and OncoGenex Technologies Inc. |
| |
|
|
|
|
| |
10.15 |
(18) |
|
OncoGenex Pharmaceuticals, Inc. Short Term Incentive Awards Program |
| |
|
|
|
|
| |
10.16 |
(18) |
|
Agreement and Consent Form (related to the Short Term Incentive Awards Program) |
| |
|
|
|
|
| |
10.17 |
(3) |
|
Form of Indemnification Agreement for Officers and Directors of the Company |
| |
|
|
|
|
| |
10.18 |
(16) |
|
Form of Indemnification Agreement between OncoGenex Technologies Inc. and each of
Scott Cormack, Stephen Anderson and Cindy Jacobs |
| |
|
|
|
|
| |
10.19 |
(16) |
|
Form of Indemnification Agreement between OncoGenex Technologies Inc. and Neil
Clendeninn |
| |
|
|
|
|
| |
10.20 |
(2) |
|
Executive Termination Agreement and General Release dated August 21, 2008 between
the Company and Michael Martino |
| |
|
|
|
|
| |
10.21 |
(2) |
|
Executive Termination Agreement and General Release dated August 21, 2008 between
the Company and Alan Fuhrman |
| |
|
|
|
|
| Exhibit |
|
|
| Number |
|
Description |
| |
|
|
|
|
| |
10.22 |
(19) |
|
Employment Agreement between OncoGenex Technologies Inc. and the Company and Scott
Cormack dated as of November 4, 2009 |
| |
|
|
|
|
| |
10.23 |
(19) |
|
Employment Agreement between OncoGenex Technologies Inc. and the Company and
Stephen Anderson dated as of November 4, 2009 |
| |
|
|
|
|
| |
10.24 |
(20) |
|
Amendment dated February 24, 2010 to the Employment Agreement between OncoGenex
Technologies Inc. and Stephen Anderson |
| |
|
|
|
|
| |
10.25 |
(19) |
|
Employment Agreement between the Company and Cindy Jacobs dated as of November 3,
2009 |
| |
|
|
|
|
| |
10.26 |
(21) |
|
Employment Agreement dated October 14, 2008 between OncoGenex Technologies Inc. and
Cameron Lawrence |
| |
|
|
|
|
| |
10.27 |
(21) |
|
Employment Amending Agreement dated January 1, 2009 between OncoGenex Technologies
Inc. and Cameron Lawrence |
| |
|
|
|
|
| |
10.28 |
(22) |
|
Securities Purchase Agreement dated as of August 15, 2005 by and among Sonus
Pharmaceuticals Inc. and the investors named therein |
| |
|
|
|
|
| |
10.29 |
(22) |
|
Form of Purchase Warrant related to the Securities Purchase Agreement |
| |
|
|
|
|
| |
10.30 |
(23) |
|
Form of Purchase Warrant issued to Schering AG |
| |
|
|
|
|
| |
10.31 |
(22) |
|
Registration Rights Agreement dated as of August 15, 2005 by and among Sonus
Pharmaceuticals Inc. and the investors named therein |
| |
|
|
|
|
| |
10.32 |
(24) |
|
Lease by and between BMR-217th Place LLC and the Company dated as of
November 21, 2006 |
| |
|
|
|
|
| |
10.33 |
(25) |
|
First Amendment to Lease by and between BMR-217th Place LLC and the
Company dated as of August 17, 2007 |
| |
|
|
|
|
| |
10.34 |
(26) |
|
Second Amendment to Lease by and between BMR-217th Place LLC and the
Company dated as
of January 28, 2008 |
| |
|
|
|
|
| |
10.35 |
(6) |
|
Amended and Restated License Agreement effective as of July 2, 2008 by and between
OncoGenex Technologies Inc. and Isis Pharmaceuticals, Inc. (OGX-011)* |
| |
|
|
|
|
| |
10.36 |
|
|
Letter Agreement Regarding Certain Sublicense Consideration for OGX-011 between
OncoGenex Technologies Inc. and Isis Pharmaceuticals, Inc. dated December 18, 2009 |
| |
|
|
|
|
| |
10.37 |
|
|
Amendment No. 1 to Amended and Restated License Agreement between OncoGenex
Technologies Inc. and Isis Pharmaceuticals, Inc. dated December 19, 2009 (OGX-011)* |
| |
|
|
|
|
| |
10.38 |
(27) |
|
License Agreement between OncoGenex Technologies Inc. and the University of British
Columbia effective as of November 1, 2001, and Amending Agreement dated as of
August 30, 2006 (OGX-011)* |
| |
|
|
|
|
| |
10.39 |
(2) |
|
Second Amending Agreement and Consent as of August 7, 2008 between the University
of British Columbia and OncoGenex Technologies Inc. (OGX-011) |
| |
|
|
|
|
| |
10.40 |
|
|
Third Amending Agreement to the License Agreement between OncoGenex Technologies
Inc and the University of British Columbia dated as of December 20, 2009 (OGX-011)* |
| |
|
|
|
|
| Exhibit |
|
|
| Number |
|
Description |
| |
|
|
|
|
| |
10.41 |
(27) |
|
Collaboration and License Agreement between OncoGenex Technologies Inc. and Isis
Pharmaceuticals, Inc. effective as of January 5, 2005 (OGX-427)* |
| |
|
|
|
|
| |
10.42 |
(27) |
|
License Agreement between OncoGenex Technologies Inc. and the University of British
Columbia effective as of April 5, 2005, and Amending Agreement dated as of
August 30, 2006 (OGX-427)* |
| |
|
|
|
|
| |
10.43 |
(2) |
|
Second Amending Agreement as of August 7, 2008 between the University of British
Columbia and OncoGenex Technologies Inc. (OGX-427) |
| |
|
|
|
|
| |
10.44 |
|
|
Collaboration and License Agreement between OncoGenex Technologies Inc. and Teva
Pharmaceutical Industries Ltd. dated as of December 20, 2009 (OGX-011)* |
| |
|
|
|
|
| |
21.1 |
|
|
Subsidiaries of the Registrant |
| |
|
|
|
|
| |
23.1 |
|
|
Consent of Ernst & Young LLP |
| |
|
|
|
|
| |
31.1 |
|
|
Certification of President and Chief Executive Officer pursuant to Rule 13a-14(a)
or 15d-14(a) of the Securities Exchange Act of 1934, as adopted pursuant to
Section 302 of the Sarbanes-Oxley Act of 2002 |
| |
|
|
|
|
| |
31.2 |
|
|
Certification of Chief Financial Officer pursuant to Rule 13a-14(a) or 15d-14(a) of
the Securities Exchange Act of 1934, as adopted pursuant to Section 302 of the
Sarbanes-Oxley Act of 2002 |
| |
|
|
|
|
| |
32.1 |
|
|
Certification of President and Chief Executive Officer pursuant to 18 U.S.C.
Section 1350, as adopted pursuant to Section 906 of the Sarbanes-Oxley Act of 2002 |
| |
|
|
|
|
| |
32.2 |
|
|
Certification of Chief Financial Officer pursuant to 18 U.S.C. Section 1350, as
adopted pursuant to Section 906 of the Sarbanes-Oxley Act of 2002 |
| |
|
|
| |
|
Schedules and similar attachments to the Arrangement Agreement have been omitted pursuant to
Item 601(b)(2) of Regulation S-K. Registrant will furnish supplementally a copy of any omitted schedule or similar attachment to the SEC upon request. |
| |
| * |
|
Confidential portions of this exhibit have been omitted and filed separately with the Commission pursuant to an application for Confidential Treatment under
Rule 24b-2 promulgated under the Securities Exchange Act of 1934, as amended. |
| |
| (1) |
|
Incorporated by reference to the Companys proxy statement on Schedule 14A filed on July 3, 2008. |
| |
| (2) |
|
Incorporated by reference to the Companys quarterly report on Form 10-Q for the quarter ended
September 30, 2008. |
| |
| (3) |
|
Incorporated by reference to the Companys Registration Statement on Form S-1, Reg. No. 33-96112. |
| |
| (4) |
|
Incorporated by reference to Companys quarterly report on Form 10-Q for the quarter ended March 31, 1999. |
| |
| (5) |
|
Incorporated by reference to the Companys current report on Form 8-K filed on March 11, 2009. |
| |
| (6) |
|
Incorporated by reference to the Companys annual report on Form 10-K for the year ended December 31, 2008. |
| |
|
|
| (7) |
|
Incorporated by reference to the Companys current report on Form 8-K filed on October 30, 2008. |
| |
| (8) |
|
Incorporated by reference to the Companys amended Form 8-A filed on July 25, 2002. |
| |
| (9) |
|
Incorporated by reference to the Companys amended Form 8-A filed on October 18, 2005. |
| |
| (10) |
|
Incorporated by reference to the Companys amended Form 8-A filed on August 14, 2006. |
| |
| (11) |
|
Incorporated by reference to the Companys current report on Form 8-K filed on May 30, 2008. |
| |
| (12) |
|
Incorporated by reference to the Companys quarterly report on Form 10-Q for the quarter ended
June 30, 2000. |
| |
| (13) |
|
Incorporated by reference to the Companys quarterly report on Form 10-Q for the quarter ended
September 30, 2006. |
| |
| (14) |
|
Incorporated by reference to the Companys proxy statement on Schedule 14A filed on April 3, 2007. |
| |
| (15) |
|
Incorporated by reference to the Companys quarterly report on Form 10-Q for the quarter ended
September 30, 2007. |
| |
| (16) |
|
Incorporated by reference to the OncoGenex Technologies Inc. registration statement on Form F-1 filed on December 13, 2006. |
| |
| (17) |
|
Incorporated by reference to the Companys registration statement on Form S-8 filed on August 26, 2008. |
| |
| (18) |
|
Incorporated by reference to the Companys current report on Form 8-K filed on April 2, 2009. |
| |
| (19) |
|
Incorporated by reference to the Companys quarterly report on Form 10-Q for the quarter ended September 30, 2009. |
| |
| (20) |
|
Incorporated by reference to the Companys current report on Form 8-K filed February 25, 2010. |
| |
| (21) |
|
Incorporated by reference to the Companys current report on Form 8-K filed March 1, 2010. |
| |
| (22) |
|
Incorporated by reference to the Companys current report on Form 8-K filed on August 18, 2005. |
| |
| (23) |
|
Incorporated by reference to the Schedule 13D filed by Schering Berlin Venture Corporation on October 31, 2005. |
| |
| (24) |
|
Incorporated by reference to the Companys annual report on Form 10-K for the year ended December 31, 2006. |
| |
| (25) |
|
Incorporated by reference to the Companys annual report on Form 10-K for the year ended December 31, 2007. |
| |
| (26) |
|
Incorporated by reference to the Companys quarterly report on Form 10-Q for the quarter ended March 31, 2008. |
| |
| (27) |
|
Incorporated by reference to the OncoGenex Technologies Inc. registration statement on Form F-1,
Amendment No. 1, filed on January 29, 2007. |